Experience Pharma Innovation at
Pharma Access at CPHI Frankfurt 2025 — Hall 9.0 · Booth G10
📅
Dates
October 28–30, 2025
📍
Venue
Messe Frankfurt, Germany
🏢
Location
Hall No. 9.0 ⎮ Booth No. G10
Discover What Sets Us Apart
At Pharma Access, we engineer state-of-the-art pharmaceutical facilities that turn innovation into reality. Here's a glimpse of what we'll be showcasing at CPHI Frankfurt 2025:
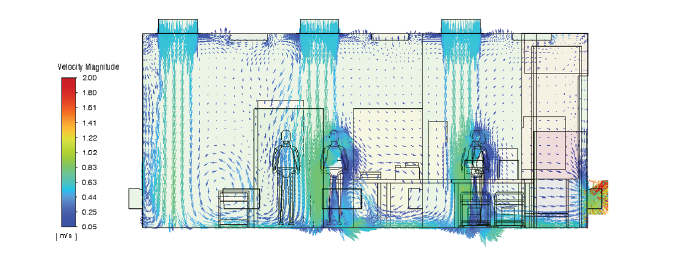
Simulation-Based Engineering
See how advanced technologies optimize pharmaceutical production with unmatched precision and efficiency.

Biotech & Pharma Compliance Focus
Explore compliance-driven solutions for mAbs, peptides, hormones, GLP-1, and sterile injectables.

Modular & Future-Ready Solutions
With MMF (Modular Mobile Facilities), step into the future of pharma construction with fully modular, rapidly deployable facilities. Scale effortlessly, reduce downtime, and bring your production online faster—without compromising quality or compliance.
Moments from Our Past Events
×
![]()
Explore Our Services