Today’s pharmaceutical projects are delivered under ever-increasing pressure. Requirements are mandated in shorter and shorter timelines. Designs are becoming more complex. Expectations from global regulatory agencies such as the US FDA and EU GMP are continuously increasing. Building the facility is just the beginning. The true test comes when you have to demonstrate that the facility will consistently operate safely and within regulatory expectations.
That’s where CQV pharma practices play an important role.
Commissioning, Qualification, and Validation bridge the gap between construction and compliant operations. Simply building a facility without a defined, lifecycle-driven CQV strategy will leave you struggling to achieve audit-ready operations.

Where Most Projects Go Off Track
Many projects still treat CQV as a phase that begins after construction is complete. At first glance, this seems logical. Build the facility, install the systems, and then validate them. But in practice, this approach often leads to delays and inefficiencies.
As systems begin testing, gaps start to appear:
- The equipment does not perform as expected
- Documentation is incomplete or inconsistent
- Integration issues emerge between systems
- Compliance requirements were not fully considered during design
At this stage, every correction becomes more expensive and time-consuming. The issue is not CQV itself. It is when and how it is introduced into the project lifecycle.
What CQV Really Means in Practice
People tend to describe CQV as three distinct tasks. In reality, CQV is a lifecycle-driven, integrated process aligned with risk-based principles. Commissioning ensures that certain systems are installed properly and functioning correctly. Qualification confirms systems meet established requirements. Validation ensures processes will reliably produce the expected result.
CQV activities work together to ensure that the facility is not just built, but is fully prepared for compliant and sustainable operations. These checkpoints have evolved with current CQV services. They are involved from the beginning of design and continue through project completion.
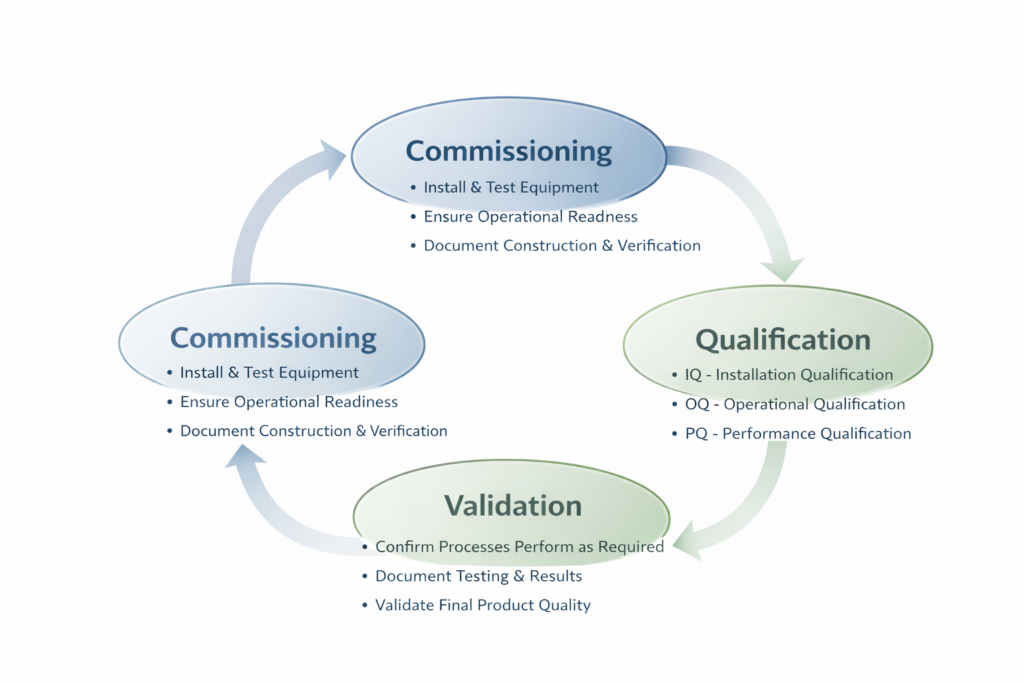
Breaking Down the CQV Lifecycle
Instead of viewing CQV as a single phase, it is more effective to understand how it evolves across the project lifecycle.
Commissioning: Proving Systems Work
Commissioning focuses on verifying that systems and equipment are installed and operating correctly.
This includes:
- Verifying installation against design specifications
- Testing system functionality
- Ensuring utilities and equipment operate as expected
At this stage, the goal is to identify and resolve issues before formal qualification begins.
Qualification: Verifying Compliance
Qualification builds on commissioning by ensuring that systems meet regulatory, user, and operational requirements under GMP conditions.
This is typically divided into stages such as:
- Installation Qualification (IQ), confirming systems are installed correctly
- Operational Qualification (OQ), verifying that systems operate within defined limits and ranges
- Performance Qualification (PQ), confirming consistent performance under actual operating conditions
Installation qualification is particularly critical, as it establishes the baseline for all subsequent validation activities.
Validation: Proving Consistent Performance
Validation focuses on process performance. It ensures that processes, utilities, and supporting systems can consistently produce results that meet predefined quality standards.
This includes:
- Process validation
- Cleaning validation
- Ongoing process verification and monitoring
Validation is what ultimately demonstrates that the facility is ready for commercial production.

Why CQV Is More Than a Compliance Requirement
There is a common perception that CQV is only about meeting regulatory expectations. In reality, it plays a much broader role in project success.
When implemented effectively, CQV:
- Reduces rework and late-stage corrections
- Improves alignment between design, construction, and operations
- Accelerates time to market
- Enhances overall project predictability and audit readiness
Well-executed qualifications and validation processes are not just about compliance. They are about ensuring operational readiness.
The Shift Toward Integrated CQV Approaches
Traditional project models treat engineering, construction, and CQV as separate activities.
This separation often leads to:
- Late identification of issues
- Misalignment between design intent and operational requirements
- Increased pressure during final project stages
Modern pharmaceutical projects are moving toward integrated CQV approaches.
In this model:
- CQV teams are involved early during design and engineering phases
- Documentation is developed in parallel with system design
- Systems are evaluated continuously, not only at the end
This reduces risk and improves overall project outcomes.

Designing with CQV in Mind
The most successful projects do not wait for CQV to begin. They design it.
This means:
- Aligning system design with qualification validation requirements from the outset
- Ensuring documentation is developed from the start
- Considering validation needs during equipment selection and layout planning
When CQV is built into the design process, the transition from construction to operation becomes significantly smoother.
How Pharma Access Approaches CQV
Here at Pharma Access, we don’t view CQV as a separate phase of a project. We incorporate it throughout the project lifecycle. By aligning engineering, execution, and CQV practices from day one, facilities are designed with qualification, validation, and compliance built in. This approach ensures stronger traceability, reduced compliance risk, minimized rework, and faster transition to validated operations.

Conclusion
CQV is often treated as the last phase of your pharma project. But it’s actually the phase that validates if your facility is ready for business.
When treated as an afterthought, it leads to delays, inefficiencies, and compliance challenges. When implemented from the beginning, it ensures speed, regulatory alignment, and operational reliability. Done as an afterthought, it creates delays & frustrations. Done right from day one, it ensures speed, compliance, and reliability.
In today’s competitive pharma market, it’s not enough to just build factories. You have to demonstrate they run compliantly, day in and day out.
That’s where CQV comes in.
FAQs
1. What is CQV in pharma?
CQV stands for Commissioning, Qualification, and Validation. It ensures that pharmaceutical facilities and systems are ready for compliant operations.
2. What is installation qualification (IQ)?
It verifies that equipment and systems are installed according to design specifications and requirements.
3. Why is CQV important in pharma projects?
It ensures compliance, reduces risks, and confirms that systems perform reliably before production begins.
4. What is the difference between qualification and validation?
Qualification verifies systems meet requirements, while validation ensures processes consistently deliver expected results.
5. When should CQV start in a project?
CQV should begin during the design phase to ensure alignment with regulatory expectations and to minimize downstream risks.
