MAGHREB Pharma 2026
Discuss your pharma facility plans in Algeria and the Maghreb region—from concept to compliant, operational delivery.
Pharmaceutical Growth in North Africa Region
Algeria's pharmaceutical industry is entering a phase of accelerated local production, supported by policy shifts aimed at reducing imports and strengthening compliance standards. This shift is driving demand for integrated project delivery models that combine engineering, construction, and validation. From facility design and cleanroom implementation to HVAC systems and commissioning support, successful execution depends on aligning with GMP expectations and local regulatory frameworks. With increasing investment across the Maghreb region, structured project planning and end-to-end delivery approaches are becoming essential for building compliant, inspection-ready pharmaceutical facilities.
What You Can Discuss with Us at MAGHREB Pharma 2026
Facility Development
- Setting up new pharmaceutical plants
- Upgrading existing manufacturing units
Engineering & Design
- Cleanroom strategy and HVAC planning
- Utilities and plant infrastructure
Project Planning
- Cost optimization and timeline planning
- Execution strategy and phasing
Compliance & Approvals
- GMP readiness and regulatory support
- Validation planning and documentation
Why Pharma Access
We integrate engineering, project delivery, and compliance into a single approach, reducing coordination gaps and overall project risk. Our focus is on building facilities that are not only operational but aligned with regulatory expectations from day one.



What Sets Us Apart
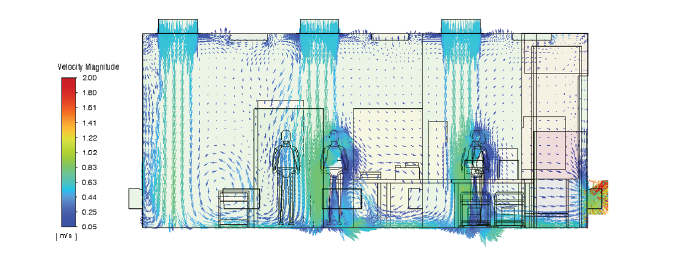
Simulation-Based Engineering
Advanced simulation tools to optimize pharmaceutical facility design with precision and efficiency.
CFD analysis, airflow visualization, particle tracking, and thermal mapping enable GMP-compliant cleanroom design. Helps identify contamination risks, optimize HVAC performance, and validate layouts before execution.

Biotech & Pharma Compliance Focus
Compliance-driven solutions for mAbs, peptides, hormones, GLP-1, and sterile injectables.
Designed as per EU GMP, US FDA, and WHO guidelines, with strong focus on contamination control strategies (CCS), cleanroom zoning, pressure cascades, and validated process flows.

Modular & Future-Ready Solutions
Modular Mobile Facilities (MMF) for scalable and rapidly deployable pharma infrastructure.
Pre-engineered modules with integrated MEP systems, cleanroom panels, and plug-and-play utilities. Enables faster installation and flexibility for expansion while ensuring GMP and Annex 1 compliance.
Engagement with Global Pharma Stakeholders
We engage with manufacturers, project stakeholders, and industry professionals across Algeria, supporting discussions on facility development, compliance, and project execution.








