Pharma projects continue to get bigger, faster, and more complex. Though engineering and construction often steal the spotlight, one aspect of pharma projects makes or breaks schedule adherence: procurement.
Procurement in today’s pharmaceutical project isn’t just about buying equipment or selecting vendors. Procurement plays a vital role in managing costs, keeping to your timeline, staying compliant with cGMP and global regulatory expectations, and realizing project success.
Pharmaceutical procurement consulting can help. Done right, procurement weaves your technical needs, vendor capabilities, regulatory compliance expectations, and project schedule into a seamless and executable plan aligned with facility engineering.
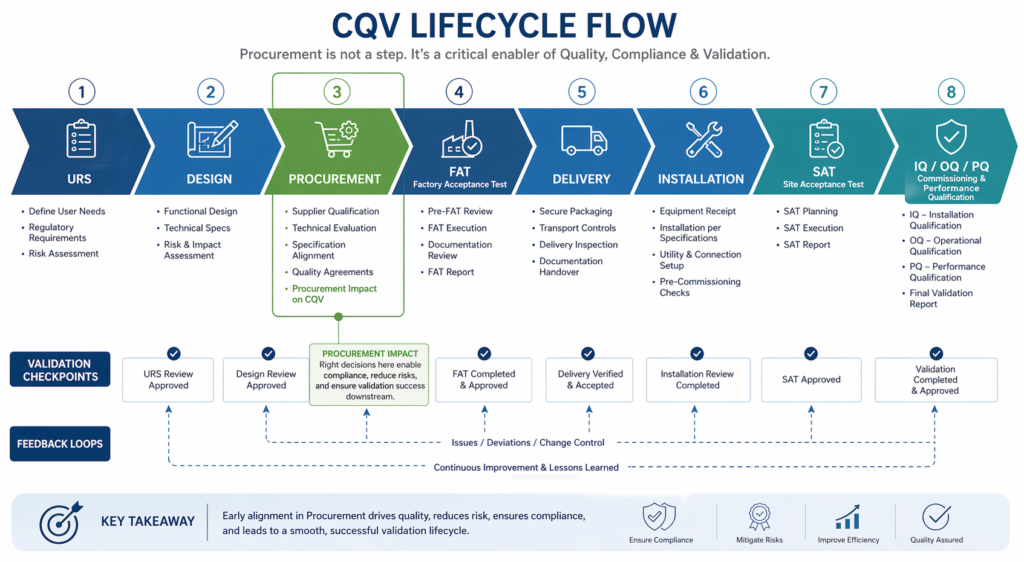
The Hidden Impact of Procurement Decisions
Procurement decisions are often made early in the project lifecycle, but their impact is felt much later during installation, integration, and qualification.
A misaligned procurement strategy can lead to:
- Delays due to late or incomplete equipment delivery
- Compatibility issues between equipment, clean utilities (PW/WFI), HVAC systems, and automation architecture.
- Increased rework during installation and commissioning and qualification
- Challenges in meeting compliance and validation requirements
These issues are rarely caused by a single decision. They are usually the result of fragmented procurement planning.
Moving Beyond Traditional Procurement Approaches
In many projects, procurement is treated as a transactional activity. Vendors are selected based on cost, availability, or previous relationships, with limited alignment to engineering and project requirements.
However, pharmaceutical projects require a more integrated approach. Modern pharma procurement consulting focuses on aligning procurement with:
- Engineering design and process requirements
- Project timelines and sequencing
- Compliance and validation strategies
- Long-term operational performance
This shift transforms procurement from a support function into a core driver of project success.
Where Procurement Strategy Creates the Most Value
The real value of procurement lies not just in cost savings, but in how effectively it supports engineering execution, compliance readiness, and lifecycle performance.
Equipment Selection with Context
Selecting equipment is not just about technical specifications. It requires understanding how the equipment fits within the broader facility design.
Key considerations include:
- Alignment with process requirements and batch sizes
- Compatibility with utilities and automation systems
- Ease of installation, operation, and maintenance
- Readiness for FAT, SAT, and downstream CQV activities
Well-informed procurement decisions ensure that equipment integrates smoothly into the facility.
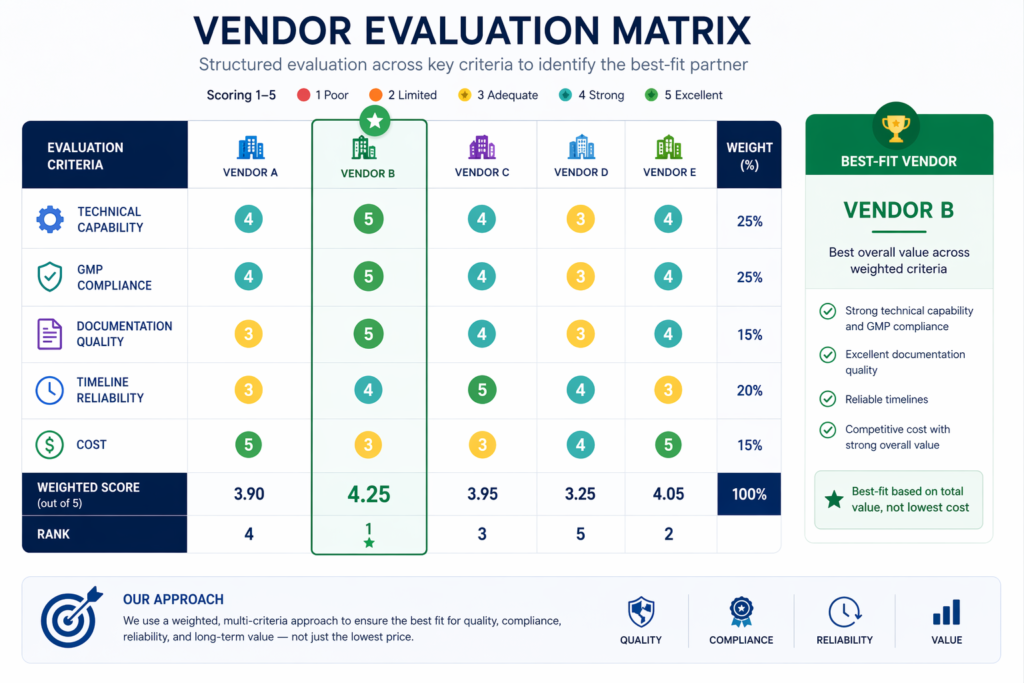
Vendor Evaluation Beyond Pricing
Choosing the right vendor is as important as selecting the right equipment. Instead of focusing solely on cost, effective procurement evaluates:
- Technical expertise and industry experience
- Ability to meet project timelines, FAT commitments, and delivery schedules
- Quality of documentation and compliance support
- After-sales service and long-term reliability
Strong vendor partnerships reduce project risk and improve execution quality, and support compliance throughout the facility lifecycle.
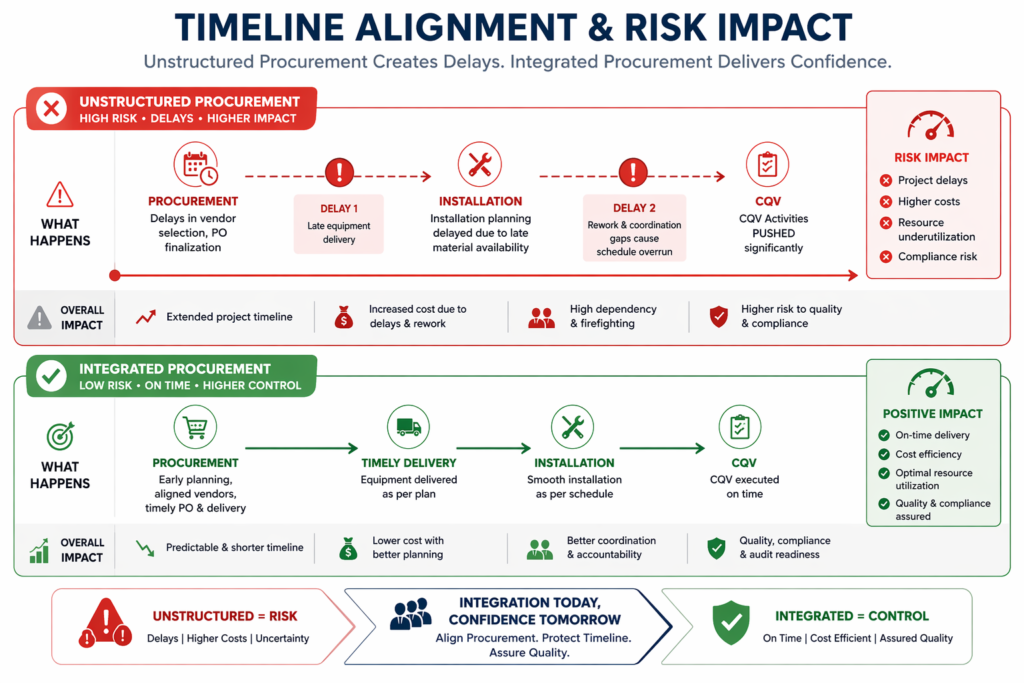
Timeline Alignment and Risk Reduction
Procurement plays a critical role in maintaining project timelines. Delays in equipment delivery or incomplete documentation can disrupt installation,commissioning and qualification schedules.
A structured procurement approach helps:
- Align delivery timelines with construction and installation phases
- Integrate FAT, SAT, and documentation review milestones into the project schedule
- Identify potential risks early
- Ensure readiness for commissioning and CQV activities
This alignment is essential for effective project management in pharmaceutical projects.
Compliance and Documentation Readiness
In pharmaceutical projects, procurement is closely linked to compliance.
Equipment and vendors must provide:
- Complete and accurate GMP-compliant documentation
- Support for validation activities including DQ, IQ, OQ, and PQ
- Compliance with regulatory standards such as cGMP, FDA, and EU GMP
Procurement decisions that overlook these aspects often lead to delays during CQV.
Procurement as Part of Project Strategy
Procurement cannot operate in isolation. It must be integrated into the overall project strategy. This requires collaboration between:
- Engineering teams defining technical requirements
- Procurement teams managing vendor selection
- Project management teams are overseeing timelines
- Quality and CQV teams ensuring compliance and validation alignment
When these functions work together, procurement becomes a driver of efficiency rather than a source of risk.
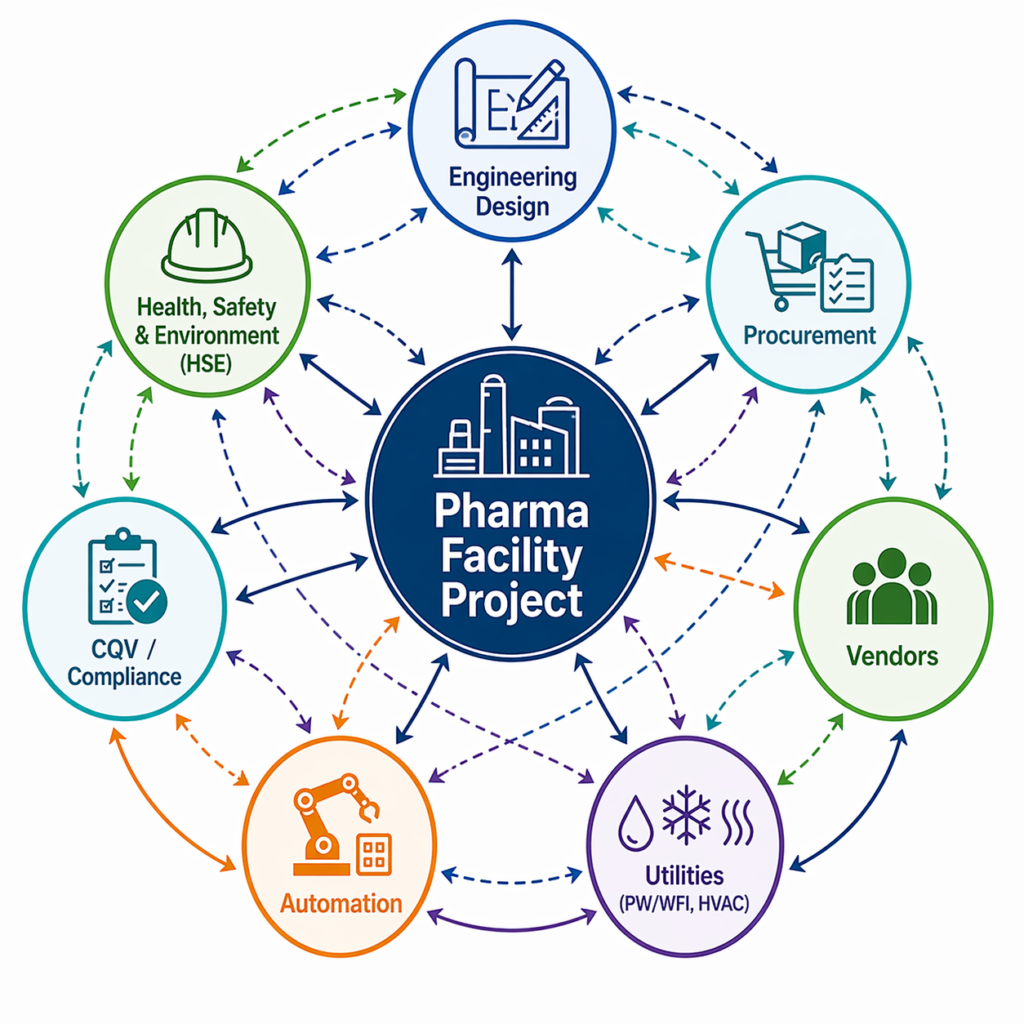
Why Fragmented Procurement Models Fall Short
Traditional procurement models often operate in silos, disconnected from engineering, CQV, and project execution. This leads to:
- Misalignment between equipment and facility design
- Reactive decision-making under timeline pressure
- Increased coordination challenges during project installation, commissioning, and qualification
As pharmaceutical projects become more complex, these limitations become more pronounced. Integrated procurement approaches are now essential to ensure seamless project delivery.

Delivering Procurement Excellence from Day One
Optimizing procurement requires a structured and proactive approach. This includes:
- Defining clear technical and compliance requirements early
- Evaluating vendors based on capability, not just cost
- Aligning procurement timelines with project schedules, FAT/SAT milestones, and CQV readiness
- Ensuring documentation and validation readiness
When procurement is planned strategically, it reduces uncertainty and improves overall project performance.
How Pharma Access Approaches Procurement Consulting
Pharma Access views procurement as an extension of of engineering and project execution, not a standalone function.
With collaborative pharmaceutical procurement consulting and sourcing services, we work to ensure equipment specifications, vendor assessments, FAT/SAT planning, and project schedules are aligned with your engineering and regulatory needs.
Leveraging deep technical expertise and execution-focused thinking, we guide clients to mitigate risk, reduce cost inefficiencies, and deliver facilities that are ready for seamless commissioning and compliant operation.
Conclusion
Procurement is often an overlooked function within pharmaceutical projects. Yet its impact can be profound.
It determines not only what gets purchased but also how quickly and successfully a facility is built, integrated, and commissioned.
Done right, procurement is a powerful enabler of efficiency, compliance, and on-time delivery.
In the new world of pharmaceutical project success, projects are not only engineered – they’re procured.
FAQs
1. What is pharmaceutical procurement consulting?
It involves optimizing equipment selection, vendor evaluation, and procurement strategies for pharmaceutical projects.
2. Why is procurement important in pharma projects?
It directly impacts cost, timelines, compliance, and project execution.
3. What factors should be considered in vendor selection?
Technical capability, experience, documentation quality, timeline reliability, and after-sales support.
4. How does procurement affect project timelines?
Delays in procurement can disrupt installation, commissioning, and qualification schedules.
5. What is the role of procurement in compliance?
Procurement ensures that equipment and vendors meet regulatory requirements and provide necessary documentation.
