Pharmaceutical manufacturing is entering an era defined by lean production, zero-defect expectations, and just-in-time delivery models. At the same time, regulatory scrutiny from agencies such as USFDA, EU GMP, and WHO continues to intensify.
Facility design is one method to help gain and maintain this delicate balance. The way a facility is laid out impacts how well the plant runs and how well it can perform to regulatory standards years down the road.
That’s where pharmaceutical facility engineering can make all the difference. The choices you make during design will impact everything else.

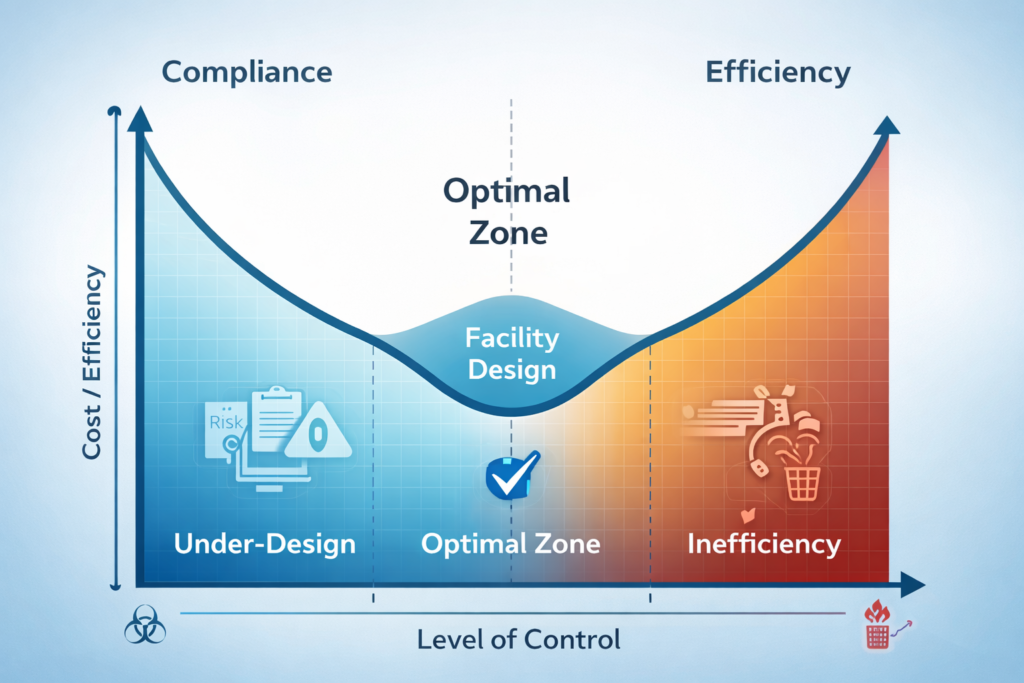
The Balancing Act: Compliance vs. Efficiency
There has long been a misconception that compliance and efficiency are at odds with one another. One requires checks, controls, and documentation aligned with cGMP guidelines. The other requires agility, throughput, and operational continuity.
But the best pharmaceutical facilities have both, built into their architecture from day one.
By designing compliance into the fabric of the facility, operational friction is minimized. By designing efficiency into the facility, the operational overhead of compliance is minimized.
The art is doing both right from the beginning.
Where Facilities Typically Fall Short
Many facilities are designed with a primary focus on meeting immediate regulatory requirements. While this ensures initial compliance, it often introduces inefficiencies that surface later during operations.
Common issues include:
- Over-engineered cleanroom spaces exceeding required ISO/GMP classifications, increasing capital and operational costs
- Complex workflows that slow down production
- Systems that meet compliance but lack flexibility
- Difficulty in scaling or adapting to new products
These are not failures of compliance. There are gaps in how design decisions are made.
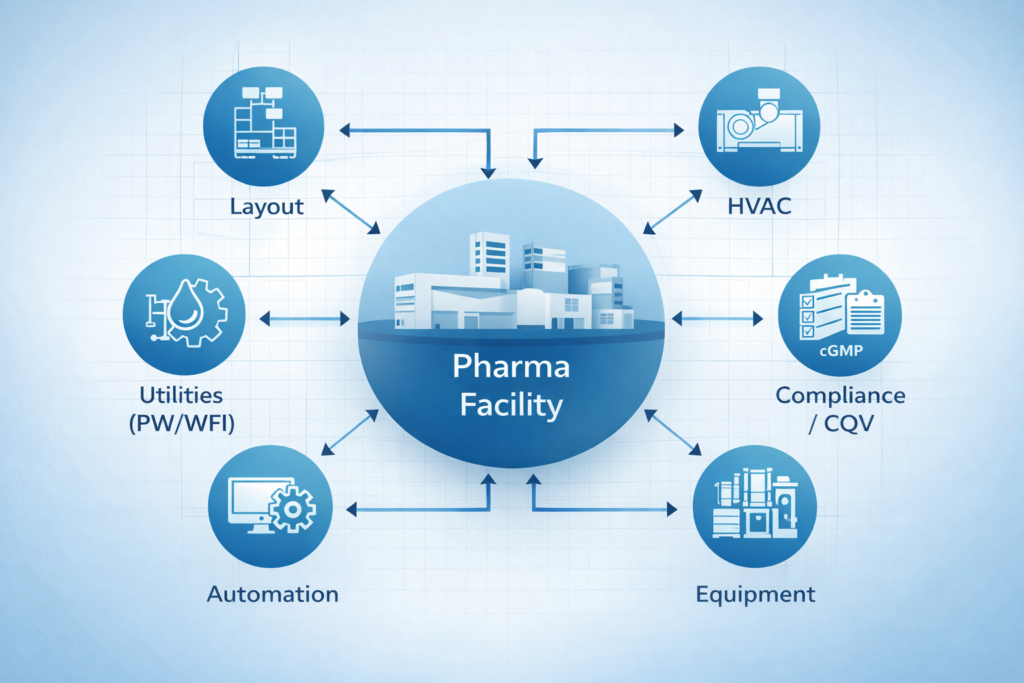
Rethinking Facility Design as a System
A pharmaceutical plant does not operate as a series of silos. It operates as a system. Layout, utilities, equipment, and processes all must work in unison.
Good pharmaceutical engineering services understand this.
We don’t design and optimize each system independently. We optimize how systems work together. This includes considering:
- Facility layout with process flow
- Utilities with equipment requirements
- Automation with operational needs
- Compliance strategy with execution
When these elements are aligned early, facilities perform more predictably and efficiently.
Design Decisions That Drive Long-Term Performance
Designing for both compliance and efficiency requires a shift in how key decisions are approached.
Layout as a Driver of Efficiency
The layout is more than a spatial arrangement. It defines how people and materials move, how processes are executed, and how contamination risks are controlled in line with GMP zoning principles.
A well-designed layout:
- Minimizes unnecessary movement
- Supports logical process sequencing
- Reduces cross-contamination risks
Poor layout decisions, on the other hand, create inefficiencies that persist throughout the lifecycle of the facility.
Utilities and Infrastructure as Enablers
Utilities such as water systems, and clean utilities are often viewed as support systems. In reality, they are critical systems that directly impact product quality, compliance, and operational stability.
Designing these systems requires:
- Alignment with process requirements
- Scalability for future expansion
- Energy-efficient configurations
When utilities are overdesigned, operating costs increase. When underdesigned, compliance risks emerge.
Equipment Integration and Flexibility
Equipment should not be selected in isolation. It must be integrated into the facility in a way that hat supports process efficiency, cleanability, and regulatory compliance.
This involves:
- Aligning equipment with process flow
- Ensuring accessibility for maintenance and cleaning
- Supporting flexibility for different batch sizes
Leading pharmaceutical plant design consultants increasingly prioritize flexible and modular approaches to equipment integration.
Automation and Data-Driven Operations
Automation is no longer optional in modern pharmaceutical facilities. It plays a key role in maintaining consistency, ensuring data integrity, and improving efficiency.
A well-designed automation strategy:
- Reduces manual intervention
- Improves process visibility
- Supports regulatory compliance
The focus is not just on implementing automation, but on integrating it effectively into the overall facility design.
Designing for Future Adaptability
One of the most important shifts in pharmaceutical facility design is the move toward adaptability.
Facilities are no longer built for a single product or process. They must support changing portfolios, new technologies, and evolving regulatory requirements.
This requires:
- Scalable infrastructure and utilities
- Flexible layouts and zoning strategies
- Modular design approaches for faster modification
Future-ready facilities are not defined by their size, but by their ability to adapt without major disruption.
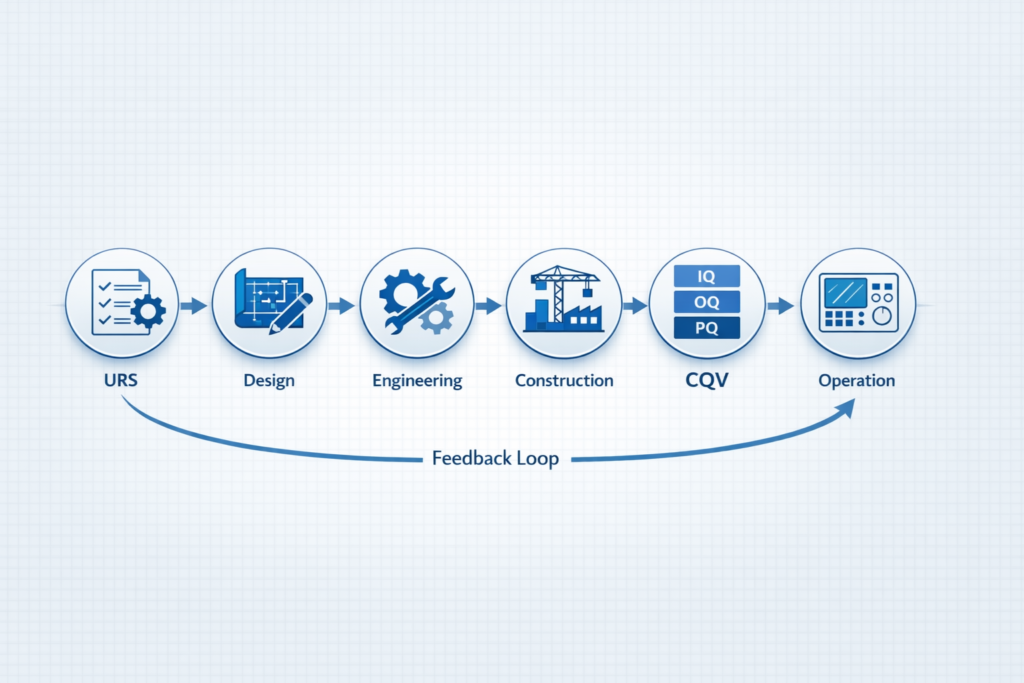
The Role of Integrated Engineering
Traditional project models often separate design, execution, and validation. This creates gaps that become visible during later stages of the project.
Integrated approaches, on the other hand, bring together engineering, quality, and execution teams from the beginning.
This alignment ensures that:
- Compliance is built into design decisions
- Systems are easier to qualify and validate
- Projects move forward with fewer delays
This is where strong pharmaceutical facility engineering capabilities create a measurable impact.
How Pharma Access Approaches Facility Design
At Pharma Access, facility design is approached as a balance between compliance, efficiency, and long-term performance, aligned with global regulatory expectations and practical execution realities.
By combining advanced pharmaceutical engineering services with execution expertise, facilities are designed to:
- Meet regulatory expectations from day one
- Operate efficiently under real conditions
- Adapt to future requirements without major redesign
This integrated approach ensures that projects are delivered with greater predictability and performance.
Conclusion
Pharma plant design isn’t just about designing to comply anymore. Today’s leaders are looking for facilities that will operate reliably, efficiently, and effectively year after year.
Exceptional facilities don’t just barely comply at the lowest cost. They build compliance into the design and efficiency into the execution.
Facility design has evolved from being a purely engineering endeavor to a business strategy that defines the pharmaceutical organization.
FAQs
1. What is pharmaceutical facility engineering?
It involves designing and developing pharmaceutical manufacturing facilities that meet regulatory (cGMP, USFDA, EU GMP) and operational requirements.
2. Why is facility design important in pharma manufacturing?
It directly impacts efficiency, compliance, scalability, and long-term performance.
3. What role do pharmaceutical plant design consultants play?
They help design facilities that balance regulatory requirements with operational efficiency and future adaptability while ensuring smooth execution and validation.
4. How can facilities achieve both compliance and efficiency?
By integrating compliance into design decisions and aligning all systems from the beginning.
5. What is the key to future-ready pharma facilities?
Flexibility, scalability, and integrated engineering ligned with evolving regulatory expectations.
