Pharma manufacturing is scaling faster than ever—but many facilities are still designed with outdated cleanroom assumptions.
As products become more complex and compliance expectations tighten, cleanroom classification is no longer just a regulatory requirement—it is a defining factor in how efficiently your facility operates.
Your cleanrooms have never just been controlled environments. They are where product quality, compliance, and operations converge.
If you want to design pharmaceutical facilities that produce consistent results without compromising compliance standards, collaborating with experienced cleanroom manufacturers and prioritizing cleanroom classification should be among the first considerations. It will affect how efficiently your facility runs, how easily it stays compliant, and how fast it can scale.

If Cleanroom Classification Is Not Considered Properly…
Projects often focus heavily on equipment and process design while underestimating the impact of cleanroom classification. However, incorrect classification decisions can create long-term operational inefficiencies. Facilities may appear compliant on paper, but struggle in real operations.
This typically results in:
- Higher operating costs due to over-designed environments
- Inefficient workflows and unnecessary restrictions
- Increased energy consumption and maintenance effort
- Delays during validation and regulatory approvals
Cleanroom classification is often treated as a compliance requirement. In reality, it is a core engineering decision that defines how efficiently a facility will operate.
Why Cleanroom Classification Matters More Than You Think
Cleanroom classifications determine the amount of particulate/microbial control necessary in different areas of the facility. Classification drives many facets of pharma engineering, such as HVAC design, material flow, personnel flow, operating procedures, etc. Classification levels that are too high result in costly facilities to operate. Too low and you risk non-compliance.
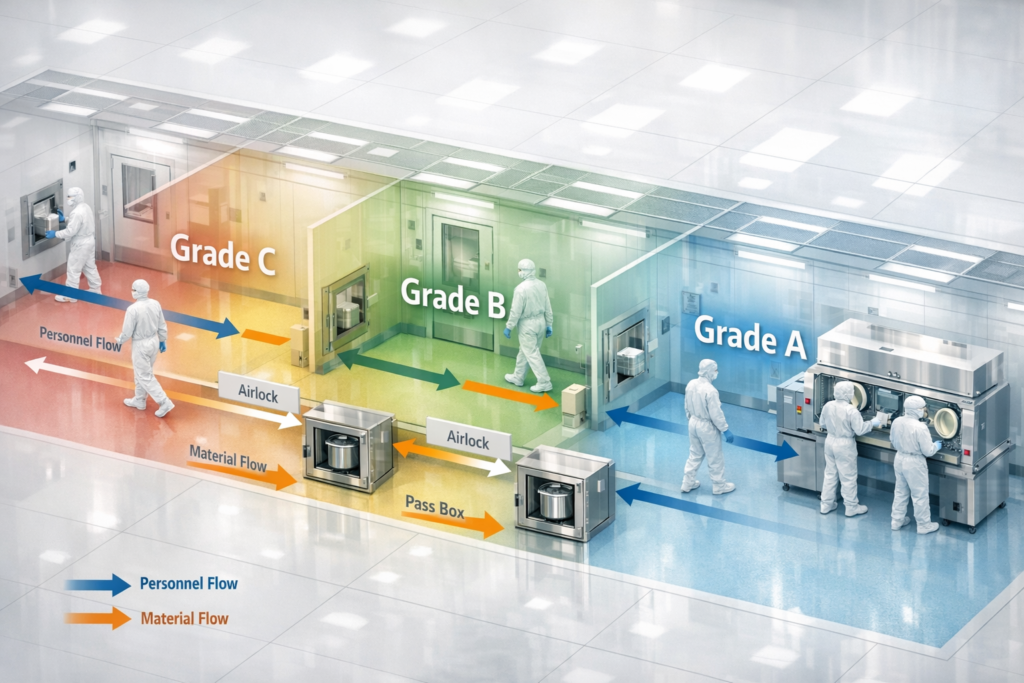
For example:
- Overclassification leads to higher HVAC loads and increased energy consumption
- Underclassification increases contamination risks and impacts quality control in the pharmaceutical industry
- Poor zoning disrupts workflow and reduces productivity
Finding the right balance is critical. Cleanroom classification should align with process needs, not assumptions.
Critical Considerations for Cleanroom Classification
While cleanroom classification is driven by regulatory requirements, its real impact is seen when translated into engineering and operational decisions.
- Aligning Classification with Process Requirements
Applying the same level of environmental control across all process steps leads to overdesign, increasing both capital investment and operational costs.
To ensure efficiency:
- Classify spaces based on actual process risk
- Avoid applying higher classifications where not required
- Align cleanroom zones with process criticality
A well-balanced classification strategy improves both compliance and efficiency.
- HVAC Design and Energy Impact
Cleanroom classification directly impacts HVAC system design.
Higher classifications require:
- Increased air changes per hour
- Advanced filtration systems
- Strict pressure control
Optimized classification reduces HVAC load while maintaining GMP compliance—making it a critical lever in pharmaceutical facility design decisions.
- Workflow and Operational Efficiency
Cleanroom classification affects how people and materials move through the facility.
Improper classification can create:
- Unnecessary movement restrictions
- Complex gowning procedures
- Bottlenecks in production flow
Efficient cleanroom design ensures smooth workflow without compromising contamination control.

- Contamination Control Strategy
Cleanroom classification is a core component of contamination control, but it cannot function in isolation.
It must be aligned with:
- Facility layout
- HVAC airflow patterns
- Cleaning and sanitization processes
In high-risk environments such as aseptic manufacturing, classification decisions become even more critical for maintaining product integrity.
- Cost vs. Compliance Balance
One of the biggest challenges is balancing compliance with cost efficiency.
Overclassification leads to:
- Higher capital investment
- Increased operational expenses
- More complex maintenance
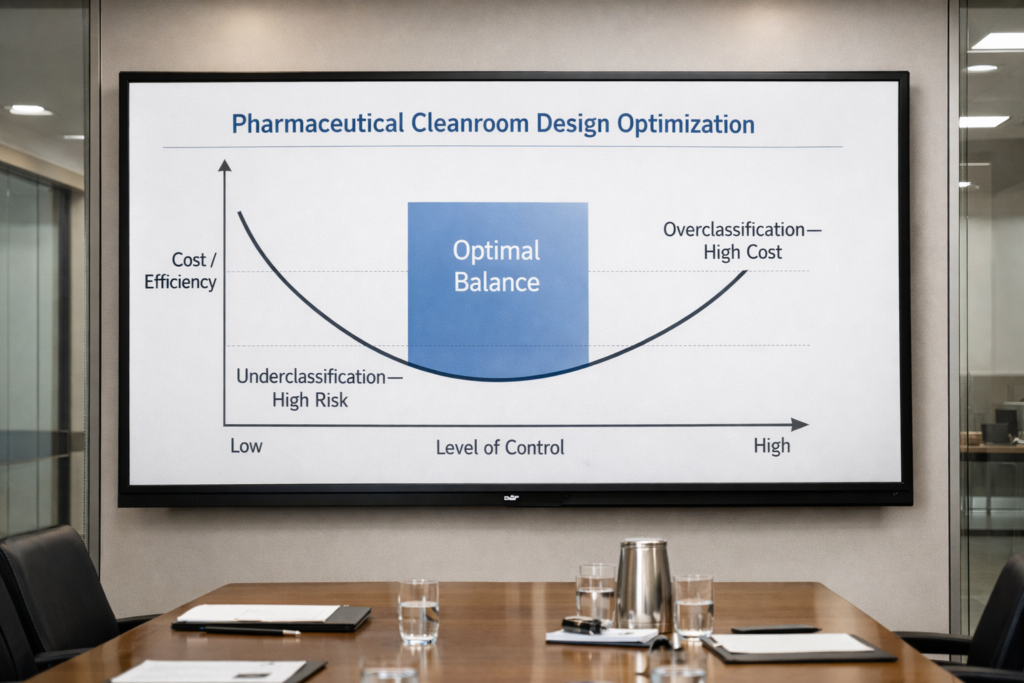
Underclassification, on the other hand, risks compliance failures. A well-designed facility finds the optimal balance between cost, compliance, and performance.
- Integration with Quality Systems
Cleanroom classification plays an important role in supporting the overall quality management system that pharma environments depend on.
It impacts:
- Monitoring systems
- Validation strategies
- Standard operating procedures
Classification decisions must align with quality systems to ensure consistency and compliance.
- Documentation and Validation
Every cleanroom classification decision must be supported by proper documentation.
This ensures:
- Clear justification of classification levels
- Alignment with regulatory expectations
- Faster validation and approval processes
Strong documentation reduces delays and improves overall project efficiency.
About Efficiency vs. Compliance
There is a common assumption that higher cleanroom classifications automatically result in better compliance. In reality, efficiency is achieved when classification levels are appropriate, not excessive.
When classification is optimized:
- HVAC systems operate more efficiently
- Operational workflows become smoother
- Maintenance and energy costs are reduced
Leading EPC companies understand that the goal is not to design the most stringent environment, but the most appropriate one.
Why Traditional Design Approaches Fall Short
In many projects, cleanroom classifications are driven by conservative assumptions rather than process requirements, leading to overdesigned, inefficient facilities.
This results in:
- Higher costs without proportional benefits
- Reduced operational flexibility
- Increased complexity in EPC construction projects
Without early integration of process, engineering, and quality teams, cleanroom classification decisions are often made in isolation.
Modern projects require integrated approaches where classification is aligned with actual process requirements and long-term operational goals.
Delivering Efficient and Compliant Cleanroom Designs
To design cleanrooms that support both compliance and efficiency, engineering teams must:
- Define classification based on process needs, not assumptions
- Integrate HVAC, layout, and workflow design early
- Balance cost, compliance, and operational efficiency
- Align the cleanroom strategy with the overall EPCM project execution
This approach ensures that facilities are not only compliant but also optimized for performance.
How Pharma Access Approaches Cleanroom Design
At Pharma Access, we see cleanroom classification as a design choice, not simply compliance-driven.
Employing Pharma Engineering best practices with seamless execution, facilities are engineered to allow you to properly balance compliance, efficiency, and scalability.
Delivering GMP-compliant facilities that “just work” for our clients day one.
Conclusion
Cleanroom classification shouldn’t just be about compliance. It should drive manufacturing efficiency.
Doing more than what is needed wastes money. Doing less than what is needed risks non-compliance. Designing correctly creates balance.
Cleanrooms that are properly classified to meet process needs and tied into the overall design strategy enable facilities to perform better, operate more efficiently, and scale with ease.
Efficient design in today’s pharmaceutical manufacturing isn’t about doing more. It’s about designing smarter.
FAQs
- What is cleanroom classification?
It defines the level of particulate and microbial control required in a controlled environment.
- Why is cleanroom classification important in pharma manufacturing?
It ensures product quality, regulatory compliance, and efficient operations.
- How does cleanroom classification impact efficiency?
It affects HVAC load, workflow, operational cost, and maintenance requirements.
- What happens if cleanroom classification is too high?
It increases energy consumption, operational cost, and complexity without added benefit.
- How is cleanroom classification linked to GMP?
It supports contamination control and is a critical part of GMP-compliant facility design.



