Why FDA Inspection Readiness Matters for Pharmaceutical Facilities
In FY2024, FDA reported 972 drug quality assurance inspections and issued 105 human drug quality-related warning letters. If you run a pharmaceutical manufacturing plant that exports to the US, a warning letter isn’t just bureaucratic red tape. Your warning letter will be posted publicly on the FDA’s website. Partners, investors, and customers will be able to read it as soon as it’s posted. Products can be held up at the border. Approvals can take months or years to revive.
The good news is that most failures are predictable. FDA Form 483 observations (the written list of findings an investigator shares with you at the end of an inspection) repeat year after year. Data integrity failures, inadequate CAPA systems, lack of process validation, and poor record keeping account for most enforcement actions. They are engineering/system failures, unavoidable surprises.
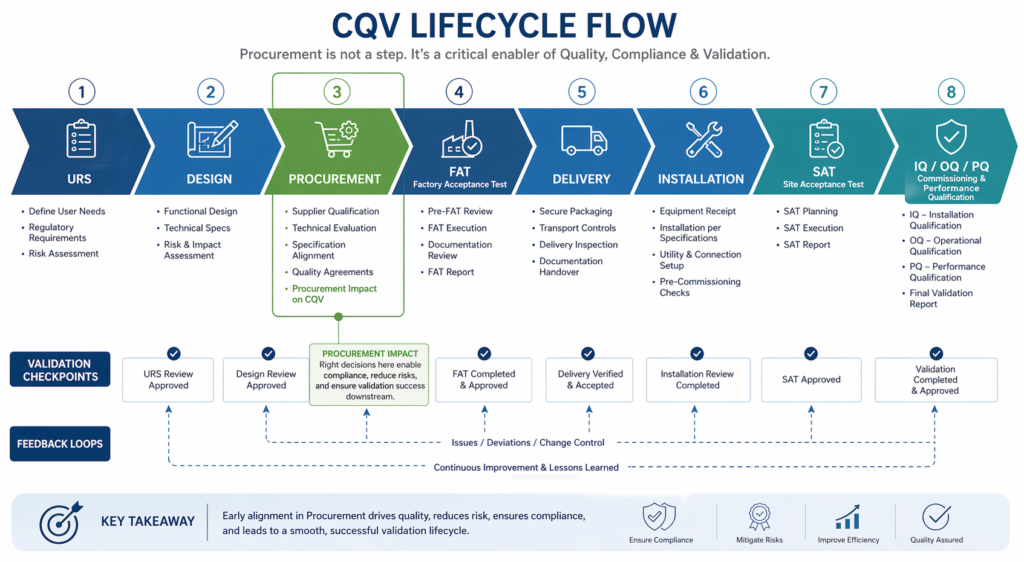
Here is your step-by-step guide to preparing your pharmaceutical manufacturing facility for a US FDA inspection, written for those who want to build inspection readiness into daily operations, not just prepare at the last minute.

Step 1: Understand What FDA Inspectors Are Actually Looking For
Before we discuss how to prepare for an inspection, let’s review what the agency is looking at. FDA inspections of pharmaceutical manufacturing facilities will primarily involve those aspects covered in 21 CFR Parts 210 and 211. Those regulations codify the current Good Manufacturing Practices for finished pharmaceuticals in the US. Common FDA inspection types include:
- Surveillance inspections – routine risk-based inspections that cover all areas including quality, production, facilities/equipment, materials, packaging/labeling, and laboratory controls
- Pre-Approval Inspections (PAI) – occur when a company submits an ANDA or NDA for a new product allowing FDA to verify that the facility can in fact manufacture the product they describe
- For-cause – inspections initiated based on complaints, adverse events, or product defects
- Follow-up inspections – conducted after a 483 or warning letter is issued to confirm the facility completed corrective actions
The FDA has increased focus on data integrity, supply chain compliance, and risk-based oversight, and in 2025 announced expanded use of unannounced inspections for foreign manufacturing facilities. Audits now include both on-site reviews and remote data verification. Plus, with the agency utilizing risk-based tools and available compliance information to identify potential areas of concern, your records should be clean and complete at all times, not just when you know a visit is coming. can be unannounced, especially for domestic facilities and increasingly for foreign facilities.
We’ll review each of these areas individually.
Step 2: Lock Down Your Documentation and Data Integrity
Documentation issues remain one of the most common and repeated areas of concern in cGMP inspections and are a chronic violation seen in pharmaceutical plant maintenance. The FDA is clear on their stance: If it wasn’t written down, it didn’t happen.
What does that mean for your facility?
Batch records are complete, accurate and traceable for every single lot produced
Laboratory data is original, unchanged, and preserved with audit trails – particularly with the use of electronic data
CAPA records clearly note root cause analysis, details of the corrective action implemented, timing, and proof that the action was effective
Written procedure deviations are reported and justified in real time
Electronic records and signatures comply with 21 CFR Part 11 validation requirements. Your systems should prevent unauthorized edits and should have full audit trails. When inspectors come, they will be looking for validation that your electronic records cannot be altered and are login restricted with tamper evident audit trails. Paper work arounds for partially automated systems are a big red flag.
If you are still using paper batch records, make validating an electronic batch record system your goal. If paper and electronic records are used together, controls should be clearly defined so there is one reliable, complete, and traceable source of truth.

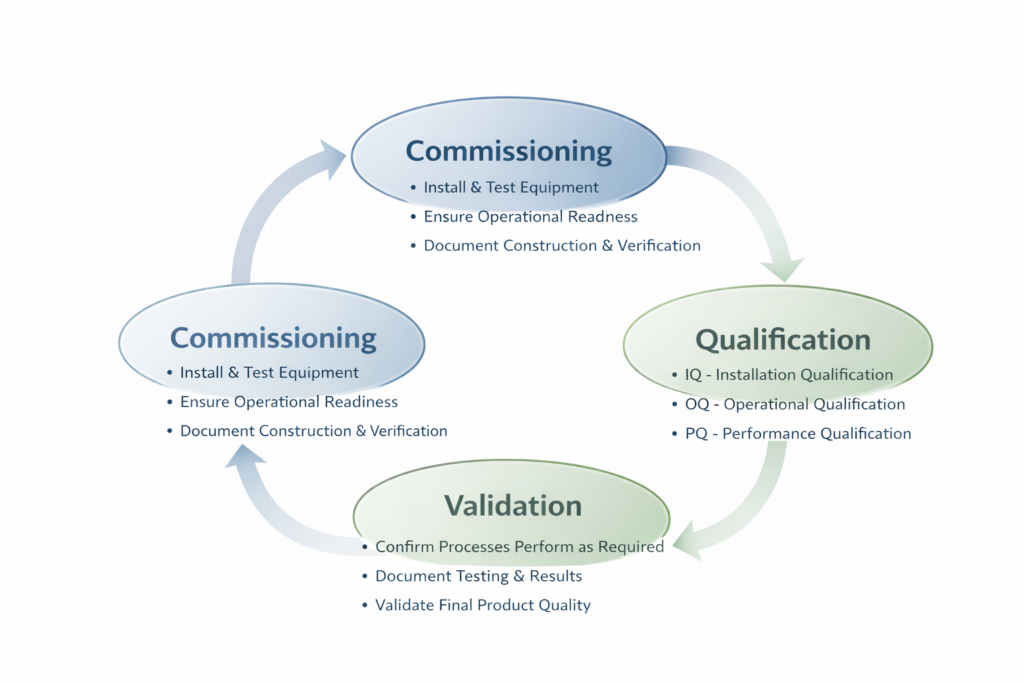
Step 3: Validate Your Facility, Equipment, and Utilities
Process validation is among the frequently observed areas in FDA inspection findings and warning letters. Validation deficiencies continue to appear in FDA enforcement trends and inspection observations.
What does validation look like in a pharma plant? Validation is the collection of documented evidence that your processes, equipment, and utilities operate within set limits. Make sure the following is validated prior to your FDA inspection:
Manufacturing processes: Process Performance Qualification (PPQ) batches should prove your process consistently manufactures products that meet all specifications. This is described in the FDA’s 2011 Process Validation Guidance as part of the process qualification stage within the lifecycle approach and is typically covered during preapproval inspections.
Cleaning processes: All equipment must be cleaned and maintained to “prevent contamination.” Per 21 CFR 211.67, your cleaning validation should prove cleaning methods reduce residues to acceptable levels AND that those levels are scientifically justified. Poorly executed cleaning validation typically results in some of the most severe 483 observations.
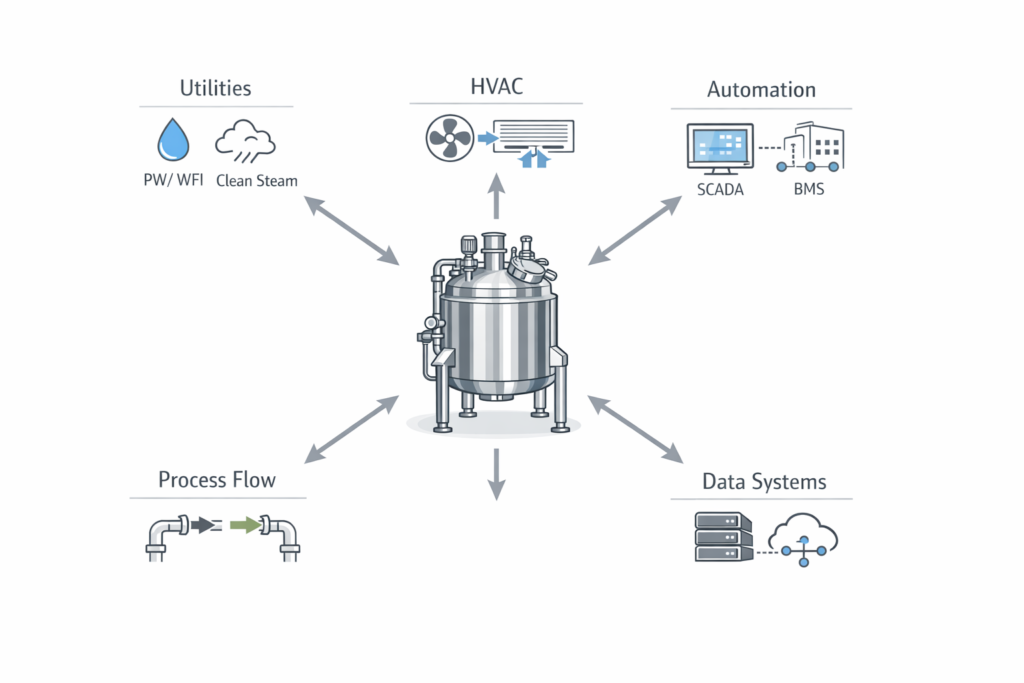
Critical utilities: WFI, clean steam, compressed air, nitrogen… all require complete qualification through IQ, OQ, PQ, or equivalent documented qualification approaches based on system risk and intended use. The FDA will verify that your utilities have been qualified, that continued monitoring is performed, and that excursions are investigated and recorded.
HVAC systems: HVAC systems should support contamination control, adequate ventilation, air filtration, temperature, humidity, and pressure control where required. Per 21 CFR 211.42 and 211.46, validation of HVAC includes qualification of HEPA filter integrity, airflow velocity, room pressure differentials, temperature/humidity control, and ISO cleanroom classification particle counts. Qualification should be performed during initial setup and reverified periodically based on cleanroom classification, risk, internal SOPs, and applicable standards. Any pharmaceutical facility that cannot produce a complete HVAC qualification package will receive observations during an inspection.
Analytical methods: Every laboratory method must be validated or verified as suitable for its intended use, depending on whether the method is compendial, non-compendial, or modified. 21 CFR Parts 211.160 through 211.194 state that method validation records should be available for all analytical methods used to assess raw materials, in-process materials, and finished goods.
This is where experienced pharma plant consultants add value. Validation planning, protocol writing, execution oversight, and summary report drafting leave room for the subject matter experts.
Step 4: Build a Functional Quality Management System
Failure modes in quality systems such as quality unit lapses, CAPA management, and deviation management continue to comprise a large percentage of FDA enforcement letters. The quality unit needs to be truly empowered. Release or rejection of a product by the quality unit must stand.
What does a working quality management system look like when your plant passes FDA inspection?
- Standard Operating Procedures are created, approved, and followed. Each procedure is version controlled and outdated procedures are not available in the plant.
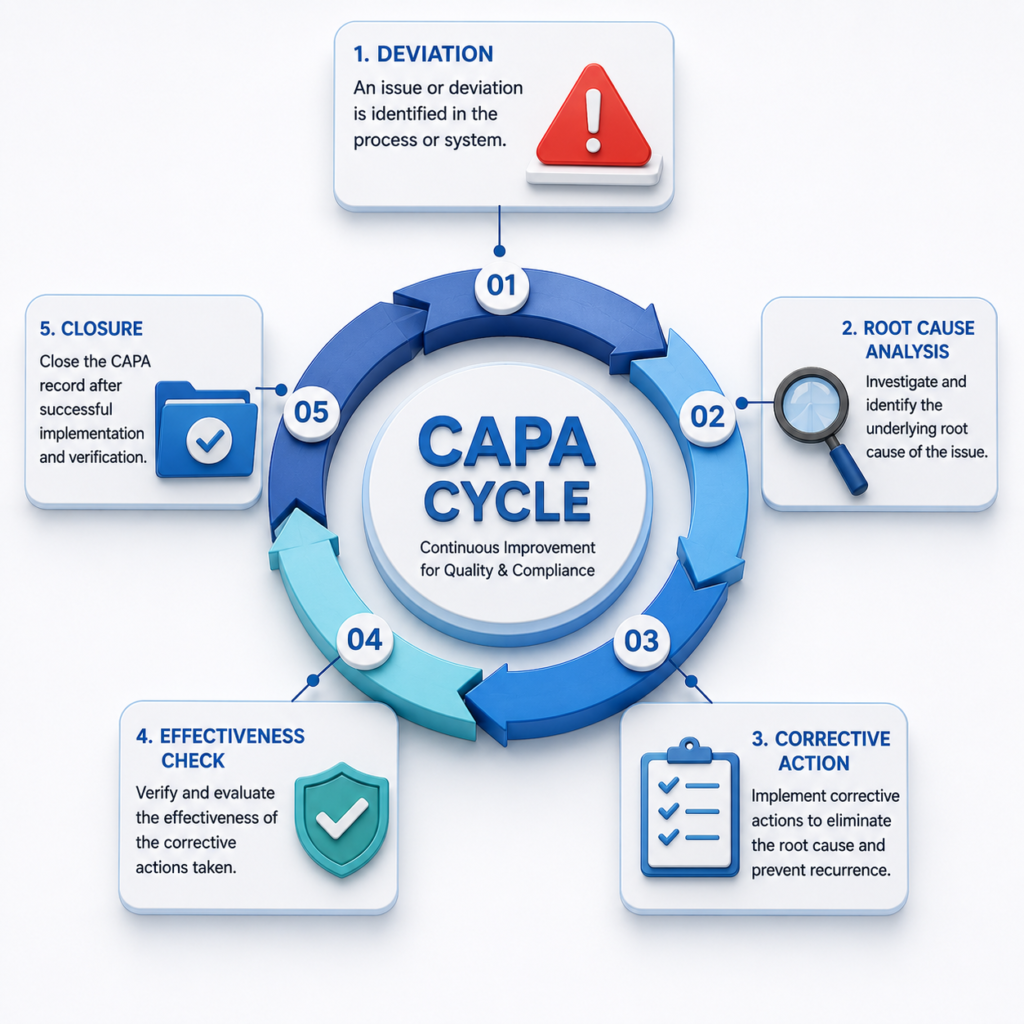
- Deviations are timely investigated, corrective actions and root causes are documented, and CAPA plans are created with defined action items, responsible parties and due dates.
- CAPAs are validated to ensure they were effective at correcting the root cause issue – not just checking a box.
- Change control includes all changes to processes, equipment, facilities or documentation and includes a risk assessment before implementation.
- Internal audits are conducted on a scheduled program and assessed by risk. Audit corrections are tracked to closure.
The FDA is now evaluating CAPA systems not only on presence but also effectiveness. A CAPA that re-opens due to failure on follow-up is a clear indication that your CAPA governance system may need stronger root cause analysis and effectiveness checks.
Step 5: Prepare Your Facility Infrastructure for Physical Inspection
Your plant’s physical condition includes buildings, facilities and equipment covered by 21 CFR Parts 211.42 through 211.68. FDA inspectors will walk around your facility. They will observe cleanroom conditions and review equipment maintenance logs, calibration tags, pest control reports, and environmental monitoring trends.
Ensure the following before any FDA inspector walks through your door:
Cleanrooms should be classified and monitored. Ensure ISO classification data, environmental monitoring reports and trend analyses are up-to-date. Document any microbial excursions and investigations.
Ensure all equipment is calibrated and maintained according to written schedules. Calibration stickers should be up-to-date. Out-of-calibration instruments found during production can lead to serious observations.
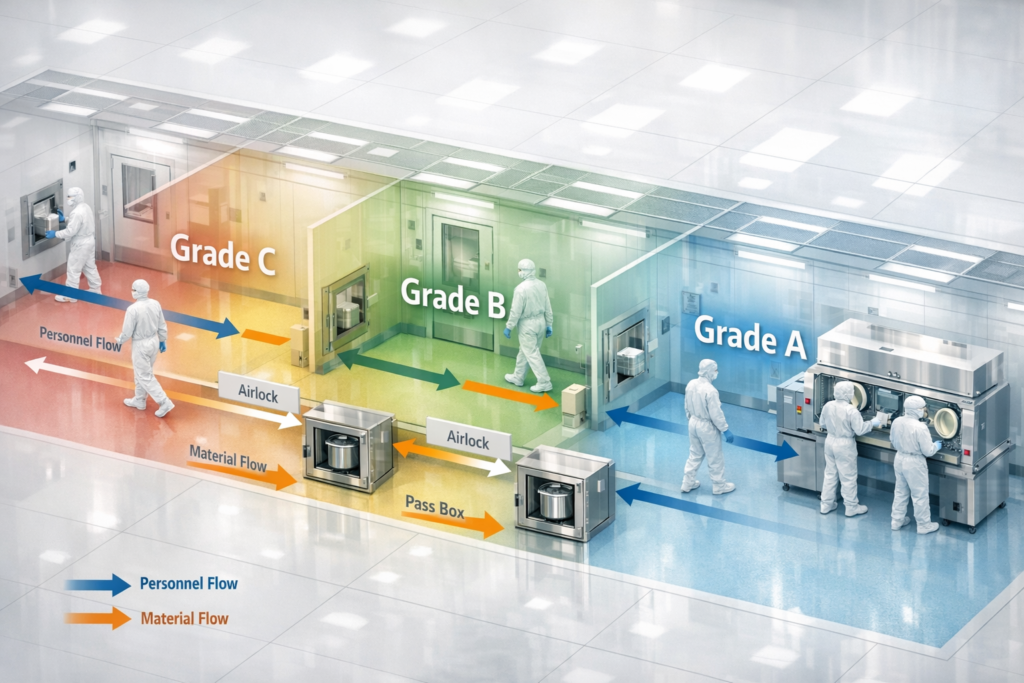
Personnel flow and material flow should prevent cross contamination. Ensure gowning procedures are written and being followed. FDA inspectors will observe gowning procedures during their facility walk-around.
Maintenance of pest control, waste disposal and facility cleaning should be documented.
Turnkey plants designed and built to cGMP from day one are much more likely to pass physical inspection than facilities converted over time through additions and upgrades. Pharmaceutical facility engineering where cleanroom layout, HVAC systems, utility routing, personnel flow and material flow are designed together produces better inspection readiness than builds constructed in steps over time.
Pharma Access, a turnkey pharma plant consulting firm with over 25+ years of experience in 120+ projects across 18 countries designs pharmaceutical facilities where CQV-focused engineering is incorporated into each stage of the project. The result: Qualification documentation becomes an expected product of the design process rather than something cobbled together at the last minute before an inspection.
Step 6: Train Your People and Run a Mock Inspection
Training is not only mandated by 21 CFR 211.25(a) where personnel must have the education, training, and experience needed to perform their assigned functions and training must be documented, it’s also an observation the FDA continually cites if staff aren’t trained. Staff that are not trained are one of the biggest causes for 483s.
However, above and beyond the regulatory requirement there are things your staff should know when the FDA shows up at your door. At a minimum, every company should:
Designate an inspection coordinator to walk the investigator around and call for documents
Prepare all department managers on potential questions the inspectors may have and how to answer them clearly and correctly
Conduct a mock inspection 6-12 months before an expected PAI window, major approval milestone, or as part of an annual inspection readiness program using the FDA’s Compliance Program Guidance Manual as your template
Correct all observations found during mock inspection
When conducting a mock inspection you will find holes in documentation, training, following through on CAPAs, and facility issues that you didn’t know existed because nobody inside the company looks at things like the FDA will.
Why the Pharma Plant Setup Matters as Much as the Preparation
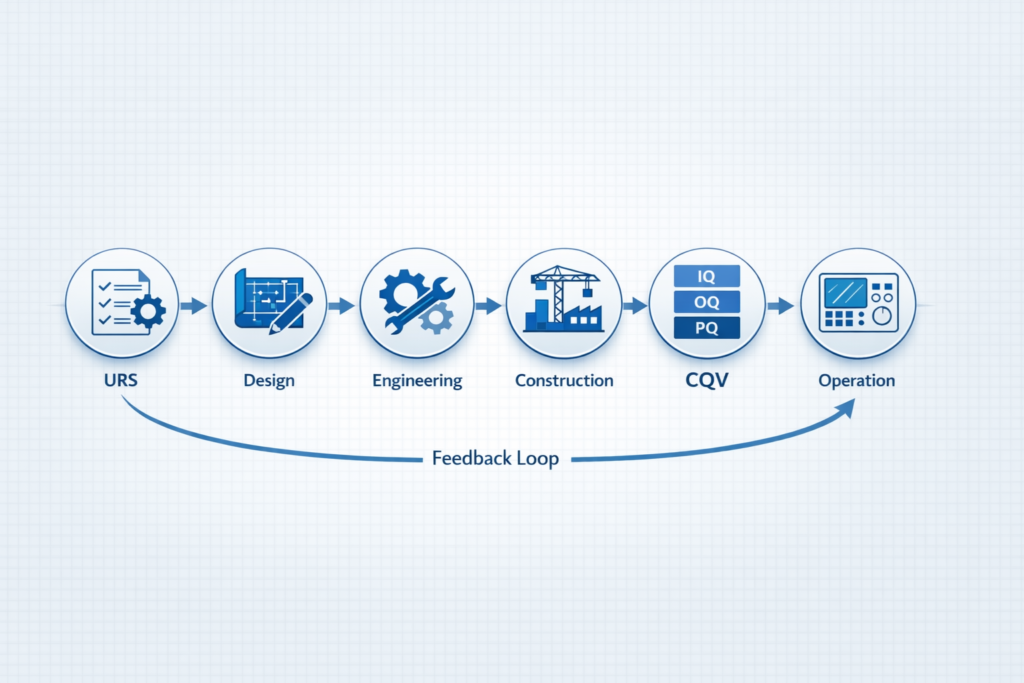
Thinking about inspection readiness should start before your plant is built. A facility designed with cGMP compliance in mind from the beginning will always be easier to inspect. Plan for HVAC qualification paths, match cleanroom classifications to product types, size and route utility systems with qualification in mind, and design material flows that prevent cross-contamination.
This is the logical reason to work with a team of pharma plant setup consultants from day one of your project. Engineering decisions and regulatory outcomes should be designed in parallel so that the qualification documentation created during construction is used as the foundation for your inspection readiness, rather than an afterthought project completed six months before the FDA walks through your doors.
Pharma Access provides complete pharma plant services from concept design through commissioning, qualification and validation. Our services cover the full range of pharma product types including sterile injectables, biotech, oral solid dosage, API, and biopharma plant operations. Visit Pharma Access to learn more about our pharma plant setup services and how we work with companies building or upgrading pharma manufacturing facilities.
FAQs
Q1: How much notice does the FDA give before a pharmaceutical plant inspection?
The FDA conducts most pharmaceutical facility inspections without advance notice. Routine surveillancemay arrive unannounced, and FDA has also announced expanded use of unannounced inspections for foreign manufacturing facilities. Pre-approval inspections may follow a drug application submission with some timing predictability, but companies should maintain inspection-ready conditions continuously rather than preparing only when an inspection is expected.
Q2: What are the most common reasons pharmaceutical plants fail FDA inspections?
The most frequent causes of FDA 483 observations and warning letters in pharmaceutical facilities are data integrity failures, weak CAPA systems, inadequate process and equipment validation, poor contamination control, and incomplete laboratory records. These issues consistently drive the majority of enforcement actions year over year.
Q3: What does a pharma plant setup consultant do to support FDA inspection readiness?
A pharma plant setup consultant helps companies design facilities to cGMP standards, develop qualification and validation protocols, author SOPs and batch record templates, perform mock inspections, and remediate gaps found during audits. The best consultants align engineering decisions with regulatory requirements from the earliest stages of facility design.
Q4: What is a Pre-Approval Inspection and when does the FDA conduct one?
A Pre-Approval Inspection (PAI) is conducted by the FDA after a company submits a new drug application (NDA) or abbreviated new drug application (ANDA). Inspectors verify that the manufacturing facility named in the application can actually produce the product as described, and that all submitted data is accurate. Facilities named for the first time in an application are prioritised for PAI.
Q5: How does facility design affect pharmaceutical plant FDA inspection outcomes?
Facilities designed with cGMP compliance as a starting requirement where HVAC qualification, cleanroom classification, material flow, and utility systems are planned together produce better inspection outcomes than converted or retrospectively upgraded buildings. A turnkey pharma plant built with CQV-focused engineering generates qualification documentation during construction that directly supports inspection readiness.