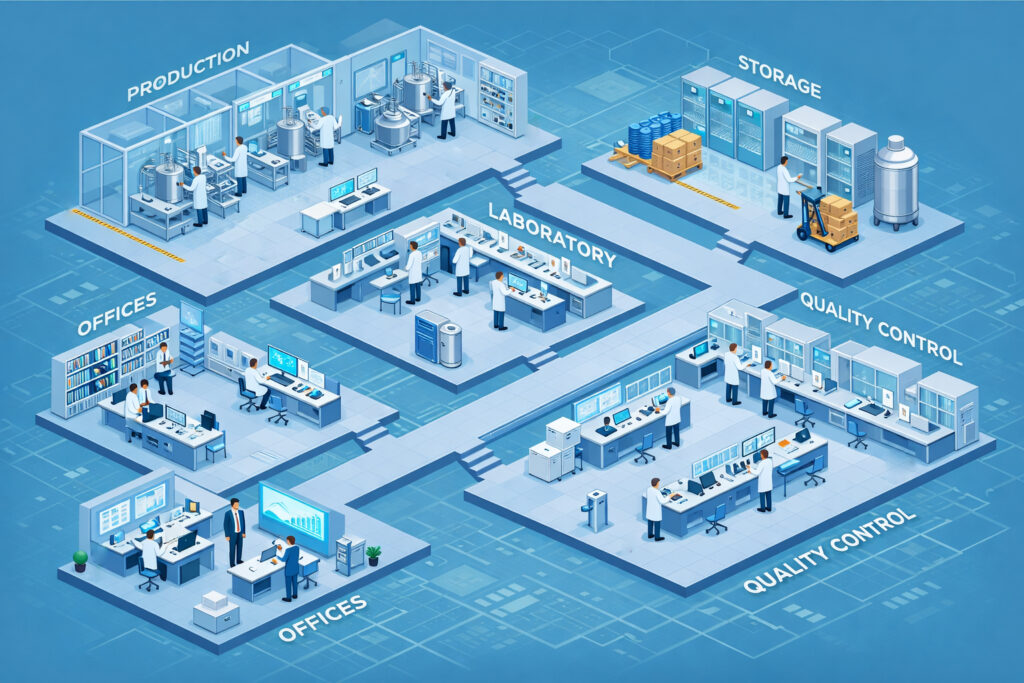
Pharma manufacturing is changing faster than ever. Therapies are becoming more complex. Product lifecycles are shrinking. Regulatory expectations are intensifying—while approval timelines continue to compress. And regulators are expecting faster approval timelines than ever. The modern pharma facility is no longer just a production space. It is a controlled environment where quality, compliance, and efficiency must work together at every step. When designing facilities that consistently manufacture quality products, compliance and speed must both be considered from the start. GMP can no longer be limited to operations and validation. It needs to be considered at the design stage itself.
When Compliance Becomes the Bottleneck
Projects today are moving quickly to build pharmaceutical manufacturing facilities. Products are expected to be commercialized faster than ever before. At the same time, regulations are becoming more rigorous. When compliance is treated as a late-stage hurdle, projects often reach commissioning phases only to discover issues that were built into the design much earlier. At that point:
- Layouts need to be reworked
- Systems require redesign
- Qualification timelines start stretching significantly
Projects are not delayed because of GMP itself—they are delayed because GMP was not integrated early enough.
Why GMP Cannot Be Retrofitted
Most EPC construction projects follow a linear approach: engineering is completed first, procurement sources materials, and construction executes the build. In many cases, compliance is only considered toward the end during commissioning and qualification. But what if GMP compliance was treated as part of the engineering scope instead of a checkbox at the end? In reality, it should be. What makes GMP unique is that once key design decisions are locked, such as facility layout, HVAC zoning, and utility routing—any modification creates cascading impact. For example:
- Changing room classification can impact airflow and pressure cascades
- Incorrect material flow may require major layout redesign
- Missing utilities can delay validation and qualification
Simply put, GMP cannot be added later. It must be built into the design from day one.
Key GMP Design Considerations
Its effectiveness depends on how well it is translated into coordinated design decisions across systems. These decisions span across multiple systems but must work together as one integrated strategy. GMP itself is a regulatory framework. It does not prescribe exact engineering solutions. However, when translated into design decisions, GMP compliance becomes more practical and structured.
- Facility Layout and Workflow
How people, materials, and waste move through a facility defines its workflow. To ensure compliance, facilities should be designed with:
- Unidirectional flow of personnel and materials wherever possible
- Clear separation between clean and non-clean areas
- Properly designed airlocks and entry/exit points
This becomes especially critical in aseptic manufacturing, where even minor contamination risks can compromise product integrity.
- HVAC and Room Environmental Design
HVAC is one of the most critical systems in a GMP facility. It maintains controlled environmental conditions required for consistent product quality. An effective HVAC design must account for:
- Room classifications
- Pressure differentials between spaces
- Airflow direction and filtration
Any instability in environmental control can directly impact quality control in the pharmaceutical industry, making HVAC one of the most critical design elements.
- Utility System Design and Clean Utilities
Utilities that come into contact with the product such as purified water (PW), water for injection (WFI), clean steam, and clean/process compressed air must be designed carefully. Key considerations include:
- Hygienic storage and distribution
- Continuous circulation to prevent stagnation
- Ease of cleaning and validation
These systems form a core part of the quality management system pharma facilities rely on to maintain consistency and compliance.
- Contamination Control Strategy
Contamination control is not driven by a single system—it is the outcome of how layout, HVAC, utilities, and operations work together. Important considerations include:
- Cross-contamination risks between products
- Interaction between personnel and material movement
- Cleaning and decontamination processes
A weak contamination control strategy can compromise an otherwise well-designed facility.
- Equipment Design and Process Flow
Equipment should not be treated as standalone assets. It must align with the overall process and facility design. Poor integration often leads to inefficiencies. Key questions to consider:
- Does the equipment align with overall process flow?
- Is it easily accessible for maintenance and cleaning?
- How well is it integrated with utilities and automation systems?
- Automation and Data Integrity Systems
Automation systems are now essential in modern pharmaceutical facilities. They not only improve operational efficiency but also support compliance. These systems support compliance by:
- Capturing and storing reliable data
- Maintaining audit trails and traceability
- Restricting user access to sensitive systems
- Documentation and Traceability
All design decisions should be traceable back to initial user requirements. Strong documentation ensures:
- Alignment between design intent and requirements
- Readiness for validation and audits
- Faster and smoother CQV processes
Documentation should not be an afterthought. It must be built into the design process.
- Designing for Compliance and Speed
There is a common misconception that GMP compliance slows projects down. In reality, projects are delayed when compliance is addressed too late. When GMP is considered from the beginning:
- Fewer design changes are required
- Rework is significantly reduced
- Commissioning and qualification timelines improve
Leading EPC companies are now integrating engineering, quality, and execution teams early in the project lifecycle. When everyone is aligned from the start, projects move faster with fewer delays.
- Where Traditional Delivery Models Fall Short
Traditional project delivery separates engineering, construction, and validation into different phases. While structured, this approach often creates gaps Common challenges include:
- Late involvement of quality teams
- Compliance efforts pushed to the end
- Decisions made without full project visibility
As projects become more complex, these gaps make compliance harder to achieve. This is why pharmaceutical projects increasingly require integrated, CQV-focused delivery approaches.
- Building GMP-Ready Facilities from Day One
Clients today expect more than just regulatory compliance. They want facilities that perform efficiently, remain compliant, and adapt to future needs. To achieve this, engineers should:
- Consider GMP at the conceptual design stage
- Align design with qualification requirements
- Involve engineering, quality, and operations teams early
- Partner with experienced pharmaceutical engineering firms
How Pharma Access Integrates GMP into Design
At Pharma Access, GMP is embedded into every stage of the project from day one. By combining precision execution with an integrated design approach, we consistently deliver facilities that meet the highest standards of compliance, efficiency, and predictability.
Final Word
GMP compliance should be addressed during the design stage, not during validation. Treating GMP as an afterthought leads to delays, rework, and inefficiencies. When considered from the beginning, it enables faster execution and better outcomes. GMP is only as strong as when it is introduced into the design process.
Frequently Asked Questions (FAQs)
1. What does GMP stand for?
Good Manufacturing Practice. It is a set of guidelines and regulations that pharmaceutical manufacturers must follow.
2. Why does GMP matter during design?
If addressed late, GMP can delay projects. When considered early, it helps maintain timelines and ensures compliance.
3. What are key GMP design considerations?
Facility layout, HVAC, utilities, contamination control, equipment integration, automation, and documentation.
4. How does HVAC impact GMP?
HVAC maintains controlled environments. Poor design can affect product safety and compliance.
5. What utilities are critical in GMP design?
Utilities such as PW, WFI, clean steam, and compressed air must be hygienic and continuously circulating.