CQV Services – Commissioning, Qualification & Validation
Change is Managed, Measured and Mastered
Managing project changes effectively ensures that scope, schedule and resource adjustments are controlled.
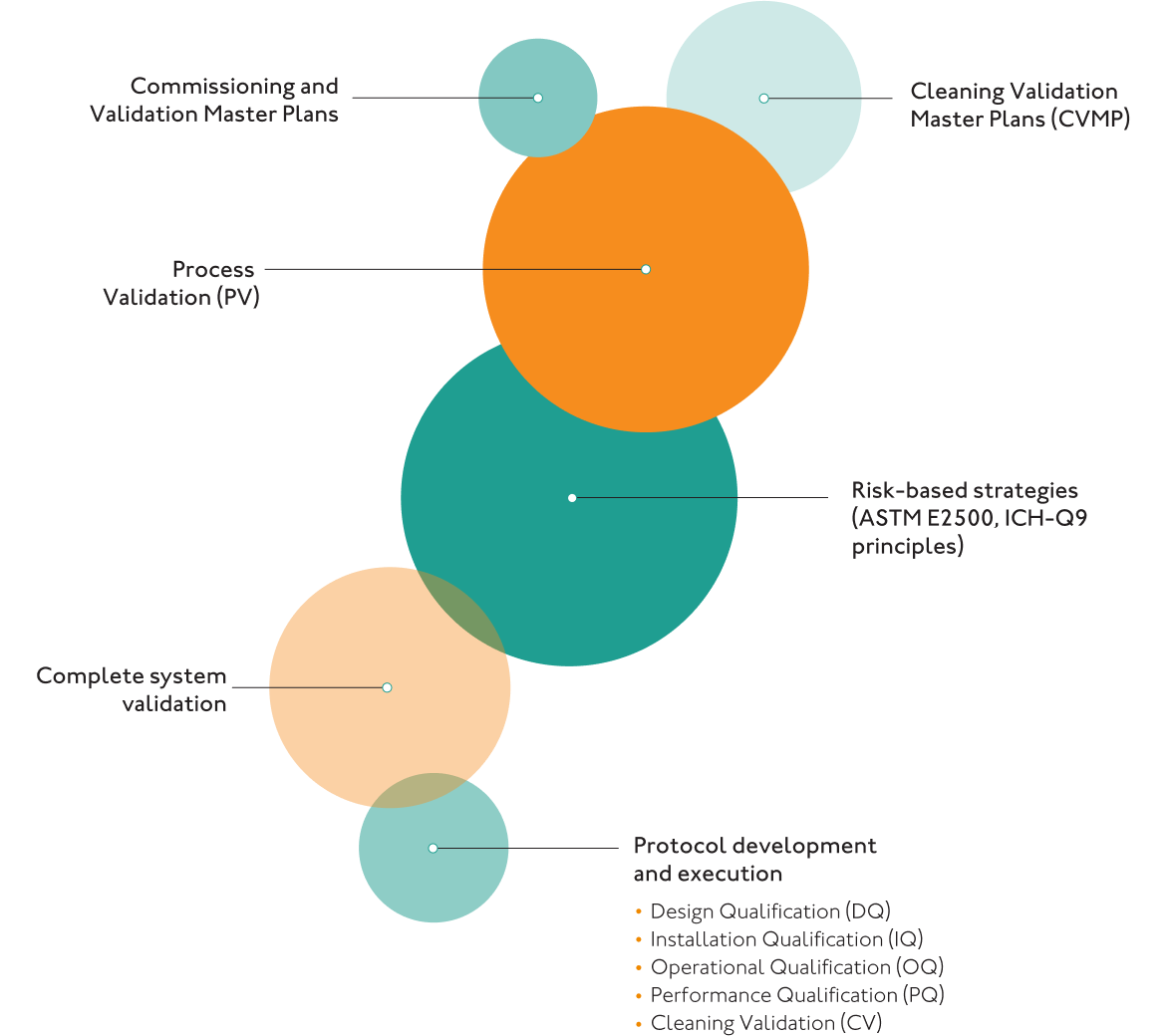
Manufacturing Process, Equipment and Machinery
- FDA 21 CFR Part 210
- FDA 21 CFR Part 211
- ISPE Baseline Guide 5 Commissioning and Qualification (Second Edition)
- ISPE GAMP 5, A Risk-Based Approach to Compliant GxP Computerized Systems
- The U.S. FDA Process Validation: General Principles and Practices
- ICH Q6A and Q6B Specifications
- ICH Q7, Good Manufacturing Practice
- ICH Q9, Quality Risk Management
- ICH Q10, Pharmaceutical Quality System
- ICH Q11, Development and Manufacture of Drug Substances
- ICH Q12, Lifecycle Management
- ASTM E 2500 Standard Guide for Specification, Design, and Verification of Pharmaceutical and Biopharmaceutical Manufacturing Systems and Equipment