Pharmaceutical facility projects are among the most complex engineering initiatives in the industrial sector. From strict regulatory expectations to highly controlled manufacturing environments, even small design errors can lead to costly delays, compliance issues, or operational inefficiencies. This is where engineering consulting services play a critical role. Experienced engineering consultants bring specialized expertise, structured project methodologies, and regulatory knowledge that significantly reduce risk across the entire facility lifecycle. For pharmaceutical companies planning new manufacturing plants, laboratory facilities, or expansion projects, working with professional engineering consultants in Mumbai and globally can help ensure projects are delivered on time, within budget, and fully compliant with regulatory standards.
In this article, we explore how engineering consultants help reduce risk in pharmaceutical facility projects and why their involvement is essential for successful project delivery.
Understanding Risk in Pharmaceutical Facility Projects
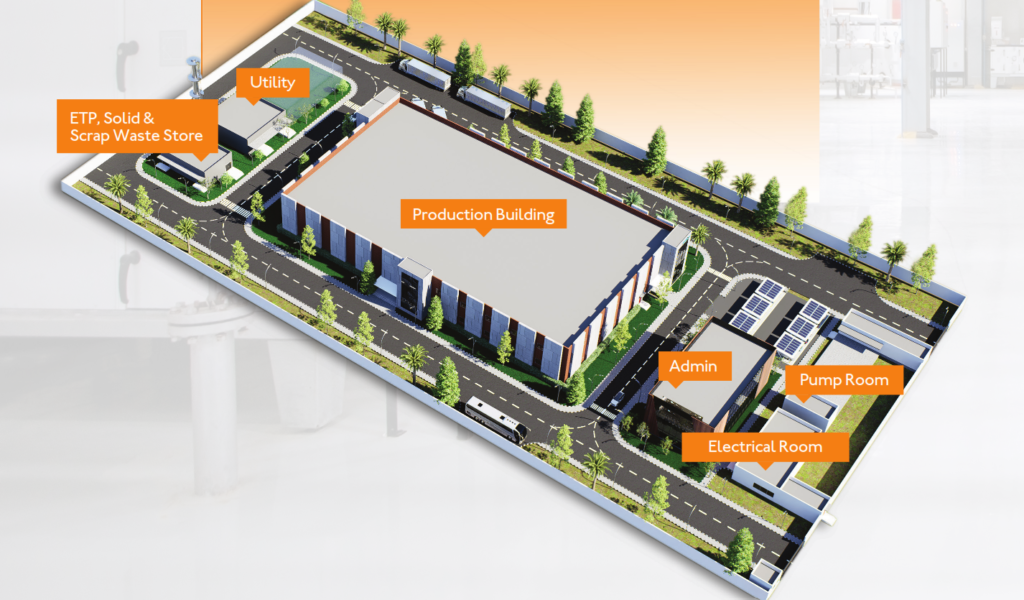
Pharmaceutical projects face several layers of risk that do not typically exist in other industrial facilities. These risks can arise during planning, design, construction, or validation phases. Common risks include:
- Regulatory compliance failures
- Facility design issues affecting product quality
- Poor integration of utilities and process systems
- Construction delays due to incomplete engineering packages
- Cost overruns due to scope gaps
- Inefficient facility layouts affecting operational performance
Engineering consultants help address these risks through structured planning, specialized design expertise, and integrated engineering coordination.
Early Engineering Planning Reduces Project Uncertainty
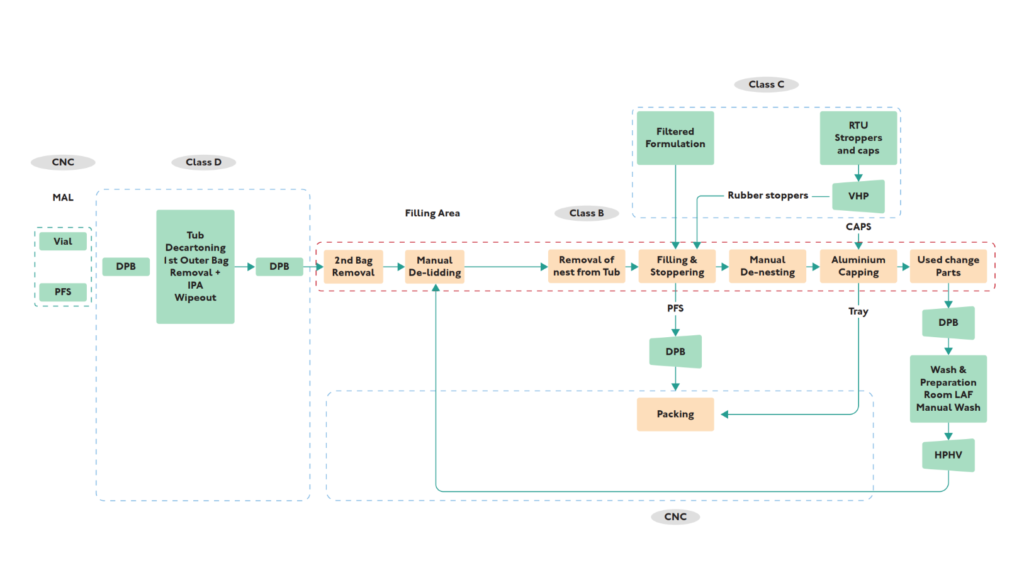
One of the most important ways engineering consulting services reduce risk is through early-stage project planning. During conceptual and front-end engineering phases, consultants help define:
- Process flow strategies
- Facility layouts and space allocation
- Material and personnel flow
- Utility demand and infrastructure requirements
- Capital expenditure estimates
This structured planning process, often referred to as front-end engineering design (FEED), provides clarity around project scope, timelines, and costs before major investments are made. Without proper front-end engineering, projects often face scope changes, budget escalation, and schedule delays later in the project lifecycle.
Regulatory Compliance Built into the Design

Pharmaceutical facilities must comply with strict global regulations such as:
- US FDA regulations (including 21 CFR Part 11)
- EU GMP Annex 1
- WHO GMP guidelines
- PIC/S standards
- ICH Q8, Q9, and Q10 (Quality by Design)
Engineering consultants also align projects with industry best practices and engineering standards such as:
- ISPE guidelines
- ASHRAE (HVAC systems)
- NFPA (fire and life safety)
- ISO standards (including ISO 14644 for cleanrooms)
To ensure compliance is built into the design, engineering consultants integrate:
- contamination control strategies
- environmental control systems
- validated utility systems
- risk-based engineering design
- Quality by Design (QbD) principles
By embedding these requirements early in the engineering process, consultants help minimize compliance risks, avoid costly redesigns, and ensure smoother regulatory inspections.
Specialized Process Engineering Expertise
Pharmaceutical manufacturing facilities depend heavily on well-designed process systems. Engineering consultants provide specialized pharma manufacturing process services, including:
- Process Flow Diagrams (PFD)
- Piping and Instrumentation Diagrams (P&ID)
- Equipment selection and sizing
- Process simulation and optimization
- automation strategy development
These deliverables ensure the facility supports safe, efficient, and compliant manufacturing operations. Without experienced engineering input, process integration issues can arise during commissioning or production, leading to operational disruptions.
Reliable Facility and Utility System Design
Utilities are the backbone of pharmaceutical manufacturing. Engineering consultants design integrated utility systems such as:
- HVAC systems for controlled environments
- Water for Injection (WFI) and purified water systems
- clean steam generation
- compressed air and process gases
- electrical distribution systems
- building management and monitoring systems
Properly engineered utility systems ensure stable facility operations and maintain the environmental conditions required for pharmaceutical manufacturing. Engineering consultants also evaluate system capacity, redundancy, and energy efficiency to ensure long-term operational reliability.
Improved Coordination Between Project Stakeholders
Pharmaceutical facility projects involve multiple stakeholders including:
- Owners and project teams
- Architects of Record (AOR)
- EPC contractors
- equipment vendors
- validation teams
Without clear coordination, projects often experience communication gaps that lead to design conflicts or construction delays. Engineering consultants help bridge these gaps by providing structured project management consulting services, coordinating engineering deliverables across disciplines and ensuring alignment between stakeholders. Their role includes:
- technical coordination with architects and contractors
- design reviews and risk assessments
- engineering documentation management
- procurement engineering support
- construction coordination
This integrated approach improves communication and helps projects progress smoothly.
Digital Engineering and Simulation Reduce Design Errors

Modern pharmaceutical engineering increasingly relies on digital engineering tools to reduce design risks. Engineering consultants use advanced tools such as:
- Building Information Modeling (BIM)
- airflow simulation for HVAC systems
- process simulation software
- electrical system modeling
- clash detection tools
These technologies allow engineers to detect design conflicts early and optimize system performance before construction begins. By identifying issues during the design phase, companies can avoid expensive changes during construction or commissioning.
Supporting Commissioning and Validation
Pharmaceutical facilities must undergo extensive validation before they can begin production. Engineering consultants support this process through:
- User Requirement Specifications (URS)
- Design Qualification (DQ)
- Installation Qualification (IQ)
- Operational Qualification (OQ)
- Performance Qualification (PQ)
Their engineering documentation ensures the facility meets regulatory expectations and supports successful validation. This reduces the risk of delays during regulatory approvals or operational start-up.
Why Engineering Consultants Are Essential for Pharmaceutical Projects
Pharmaceutical projects require deep expertise in process engineering, facility design, utilities, and regulatory compliance. Professional engineering consulting services bring this expertise together, helping pharmaceutical companies manage complexity and reduce risk throughout the project lifecycle. By working with experienced engineering consultants in Mumbai and internationally, companies gain access to specialized knowledge, proven engineering methodologies, and integrated project coordination. Combined with strong project management consulting services, engineering consultants help ensure pharmaceutical facilities are designed for compliance, operational efficiency, and long-term reliability.
Conclusion
Developing pharmaceutical manufacturing facilities is a high-stakes investment that requires precise engineering and careful risk management. Engineering consultants play a critical role in minimizing project risk by providing structured planning, specialized design expertise, regulatory compliance integration, and stakeholder coordination. From concept design to validation readiness, engineering consulting partners help pharmaceutical companies deliver facilities that meet regulatory expectations while supporting efficient and scalable manufacturing operations. As pharmaceutical manufacturing continues to evolve, the role of experienced engineering consultants will remain essential in delivering safe, compliant, and future-ready facilities.



