Making medicine is no longer just about scale. It’s about delivering flexibility, precision, and consistency while adhering to increasingly stringent cGMP and global regulatory requirements. A lot rides on one decision: pharmaceutical process equipment selection. Equipment isn’t just there to perform a process. It impacts product quality, operating efficiencies, readiness to comply, and future scalability. The wrong equipment can bake inefficiencies into your facility for years to come. Equipment selection should be treated as a core engineering and design decision, not a downstream procurement activity.
When Equipment Selection Goes Wrong
In many projects, equipment is selected based on initial cost, vendor familiarity, or isolated process requirements. While this may seem efficient in the short term, it often creates long-term challenges across operations, validation, and compliance.
Facilities may experience:
- Frequent process interruptions due to mismatched equipment capacity
- Difficulties in cleaning, validation, and maintenance
- Integration challenges with utilities and automation systems
- Reduced operational flexibility when product requirements change
These issues are not always visible during installation. They emerge during operations, when the cost of correction is significantly higher.
Rethinking Equipment Selection in Modern Pharma Facilities
Effective design of modern facilities requires engineered equipment selection aligned with process and regulatory intent, not just adding more assets. Equipment selection needs to consider project goals around efficiency, compliance and flexibility.
That includes how the equipment works within the context of:
- Process flow
- Plant layout
- Utilities
- Automation/data integrity
- Future manufacturing capacity
Equipment selection should be part of your overall pharma engineering strategy, not a separate task.

Key Factors That Define the Right Equipment Choice
Selecting the right pharma manufacturing equipment requires balancing multiple considerations. These factors must work together to support both immediate production needs and long-term operational goals.
- Process Compatibility and Performance
The first and most fundamental question is whether the equipment aligns with the intended process.
Equipment should:
- Support required batch sizes and process conditions
- Maintain consistency and repeatability
- Deliver reliable performance under varying production demands
Mismatch at this level often leads to inefficiencies that cannot be corrected through adjustments later.

- Flexibility for Changing Production Needs
Pharmaceutical manufacturing is becoming increasingly dynamic especially with the rise of multi-product facilities and biologics.. Facilities must be able to adapt to new products, varying batch sizes, and evolving market demand.
Flexible equipment allows:
- Faster changeovers between products
- Scalability without major capital investment
- Reduced downtime during transitions
Rigid systems may meet current requirements but limit future growth.
- Ease of Cleaning and Validation
In regulated environments, equipment must be designed for compliance as much as performance.
Key considerations include:
- Cleanability and hygienic design
- Compatibility with cleaning and sterilization processes
- Ease of validation and documentation
Equipment that is difficult to clean or validate can slow down operations and increase compliance risk and increase compliance risk under GMP guidelines.
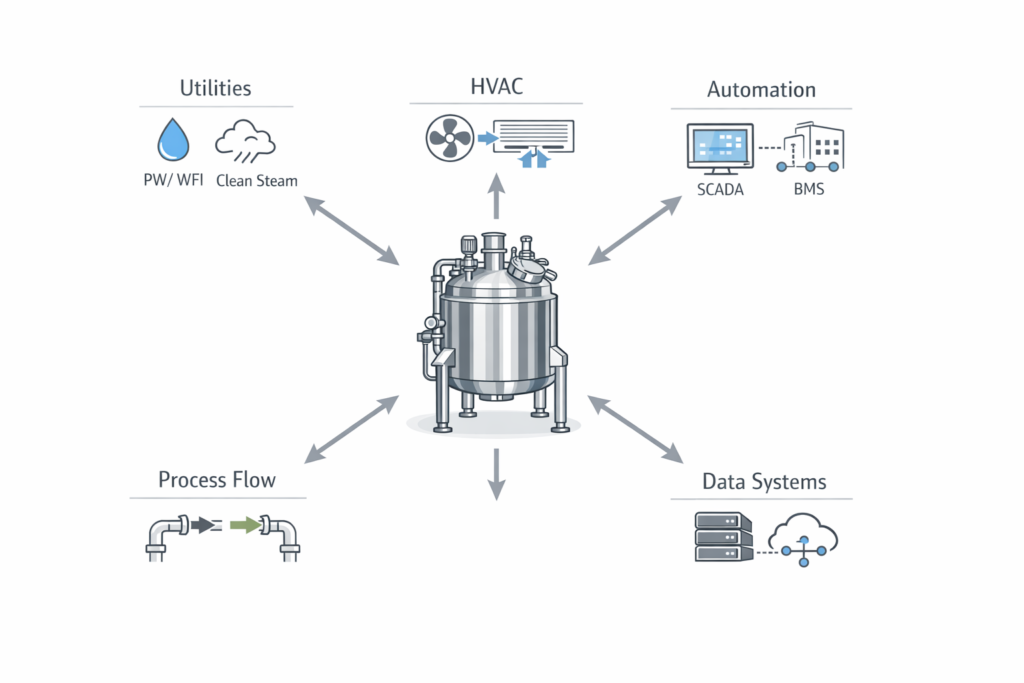
- Integration with Facility and Utilities
Equipment does not operate independently. It must integrate seamlessly with the overall facility.
This includes:
- Alignment with process flow and layout
- Compatibility with utilities such as water, steam, and compressed air
- Integration with automation and control systems
Poor integration often leads to operational inefficiencies and increased project complexity.
- Automation and Data Integrity
Modern pharmaceutical facilities rely heavily on automation for both efficiency and compliance.
Equipment should support:
- Data capture and monitoring
- Integration with SCADA, BMS, and other systems
- Audit trails and traceability
This ensures alignment with regulatory expectations and supports consistent operations.
- Lifecycle Cost, Not Just Capital Cost
Focusing only on upfront cost often leads to higher expenses over the lifecycle of the facility.
A better approach considers:
- Maintenance requirements
- Energy consumption
- Downtime and reliability
- Spare parts availability
The goal is to select equipment that delivers value across its entire lifecycle.
- Vendor Capability and Support
Equipment performance is also influenced by the capability of the vendor.
Important factors include:
- Technical expertise and industry experience
- After-sales support and service
- Availability of documentation and compliance support
Strong vendor partnerships reduce risk and improve long-term reliability.

Equipment Selection and Manufacturing Efficiency
The right equipment selection has a direct impact on manufacturing efficiency.
When aligned correctly:
- Processes run more consistently
- Downtime is minimized
- Changeovers are faster
- Compliance requirements are easier to maintain
Efficiency is not achieved by adding more equipment. It is achieved by selecting the right equipment and integrating it effectively.
Why Traditional Selection Approaches Fall Short
Traditional approaches often treat equipment selection as a procurement-driven activity.
This leads to:
- Decisions made without full visibility of the process and facility requirements
- Limited coordination between engineering and operations teams
- Challenges during installation, integration, and validation
As pharmaceutical projects become more complex, this approach is no longer sufficient. Integrated, engineering-led selection processes are essential for ensuring that equipment supports both compliance and efficiency.
Delivering the Right Equipment Strategy from Day One
To ensure optimal outcomes, equipment selection must be approached strategically.
Engineering teams should:
- Evaluate equipment within the context of the full facility design
- Align selection with process, utilities, and automation systems
- Consider long-term operational and compliance requirements
- Involve stakeholders across engineering, quality, and operations early
This approach reduces risk and ensures smoother project execution.
How Pharma Access Supports Equipment Selection
Equipment selection at Pharma Access is never made in isolation. It is part of the overall project plan. By combining pharma engineering expertise with execution experience, we ensure that process equipment aligns with process requirements, facility design, and global regulatory expectations. Helping clients realize efficient, scalable, and future-proof manufacturing.

Conclusion
Choosing appropriate equipment is one of the most critical decisions during facility design. The impact reaches far beyond day-to-day operations and into overall facility performance. Strategic equipment selection and integration enable efficiency, compliance, and scalability. Success in today’s pharmaceutical manufacturing is measured by quality, not by how much equipment you have.
FAQs
1. What is pharmaceutical process equipment?
It refers to equipment used in manufacturing processes such as mixing, granulation, filtration, and packaging.
2. Why is equipment selection important in pharma manufacturing?
It directly impacts efficiency, product quality, compliance, and operational flexibility.
3. What factors should be considered when selecting equipment?
Process compatibility, flexibility, cleanability, integration, automation, lifecycle cost, and vendor support.
4. How does equipment impact manufacturing efficiency?
The right equipment reduces downtime, improves consistency, and enables faster changeovers.
5. What is the biggest mistake in equipment selection?
Focusing only on initial cost instead of long-term performance and integration.



