Pharma facilities are precision factories. Critical systems, connections, and flow paths must all function predictably in highly regulated environments.
Process piping is often the unsung hero of those systems. It’s quietly responsible for product quality, safe operation, and regulatory compliance.
Today’s pharmaceutical process piping systems not only transport fluids, but they also help maintain fluid purity and prevent cross-contamination. They help ensure batch-to-batch process consistency by supporting hygienic flow, cleanability, and reliable utility distribution.
Done right, piping allows processes to flow. Done wrong, piping can create risk, headaches, and non-compliance.
Why Piping Design Deserves More Attention
Process piping is possibly the most interconnected system in a pharmaceutical building. It connects utilities to equipment, points of use, instrumentation, drains, and process systems. .
Ironically, piping design is one of the last things that gets done. Many times, it’s an afterthought once equipment and rough layouts have been defined.
This approach can lead to:
- Dead legs that promote microbial growth
- Difficult-to-clean systems
- Pressure drops and flow inconsistencies
- Delays during commissioning, qualification, and validation
These are not minor issues. They directly impact product quality and regulatory compliance.
Beyond Flow: What Piping Design Really Controls
Piping systems do more than move fluids. They influence how processes perform and how easily systems can be maintained and validated.
Effective pharmaceutical engineering design considers piping as a critical control system rather than a supporting utility.
Well-designed piping ensures:
- Hygienic flow without contamination risks
- Consistent delivery of process fluids
- Ease of cleaning and sterilization
- Reliable system performance over time
- Better accessibility for maintenance and inspection
In regulated environments, these factors are essential for maintaining compliance.
Designing for Hygiene and Cleanability
One of the most important aspects of piping design is ensuring hygienic conditions.
This requires attention to details that are often overlooked.
For example:
- Avoiding dead legs where fluids can stagnate
- Maintaining a proper slope for drainage
- Using appropriate materials and surface finishes
- Designing for clean-in-place (CIP) and sterilize-in-place (SIP) systems
- Ensuring proper valve orientation, low-point drains, venting, and minimal product hold-up
Even small design oversights can create areas where contamination risks increase.
Material Selection and System Integrity
The materials used in piping systems must meet both process and regulatory requirements.
Common considerations include:
- Corrosion resistance
- Compatibility with process fluids
- Surface finish to support cleanability and reduce microbial retention risk
- Suitability of gaskets, elastomers, valves, welds, and fittings
Material selection is not just a technical decision. It directly impacts system longevity and compliance.
Integration with Equipment and Utilities
Piping systems must align with equipment and utility systems across the facility.
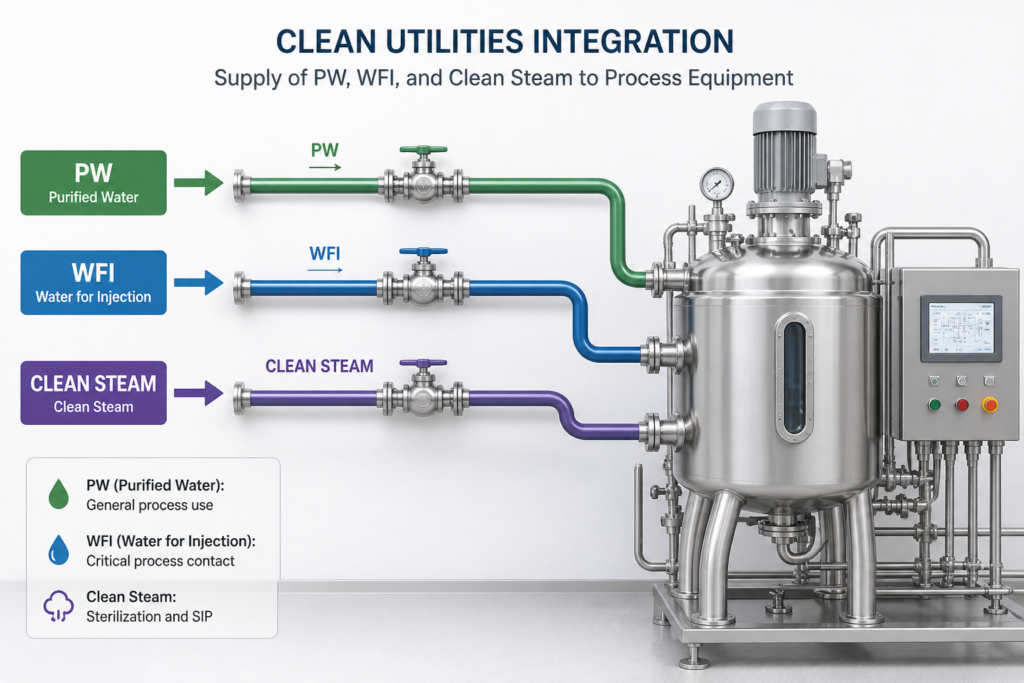
This includes:
- Connecting clean utilities such as PW, WFI, and clean steam
- Ensuring proper routing to support process flow
- Avoiding congestion and accessibility issues
- Coordinating with HVAC, automation, electrical, equipment layout, and CQV requirements
Poor integration can lead to installation challenges and operational inefficiencies.
Designing for Validation and Compliance
In pharmaceutical environments, GMP-impacting systems must be appropriately commissioned, qualified, and, where required, validated.
Piping design plays a key role in ensuring that validation is achievable and efficient.
This involves:
- Ensuring traceability of materials and components
- Supporting testing and inspection activities
- Providing clear documentation for qualification
- Maintaining weld records, pressure test documentation, slope verification, and accurate as-built drawings
Strong alignment between piping design and validation requirements reduces delays during CQV.
Safety Considerations in Piping Design
Safety is a critical aspect of piping design, especially when handling high-temperature fluids, pressurized systems, or hazardous materials.
Key safety aspects include:
- Pressure control and system integrity
- Leak prevention and detection
- Proper insulation and protection
- Emergency shutdown and isolation mechanisms
A well-designed piping system protects both the product and the people operating the facility.
The Cost of Poor Piping Design
Piping issues are often discovered late during commissioning or operation.
At that stage, corrections become complex and expensive.
Common consequences include:
- Rework during installation
- Delays in qualification
- Increased maintenance requirements
- Risk of compliance deviations
- Reduced cleanability, accessibility challenges, and avoidable operational downtime
Investing in proper design early reduces these risks significantly and supports faster, smoother project delivery .
Why Integrated Engineering Matters
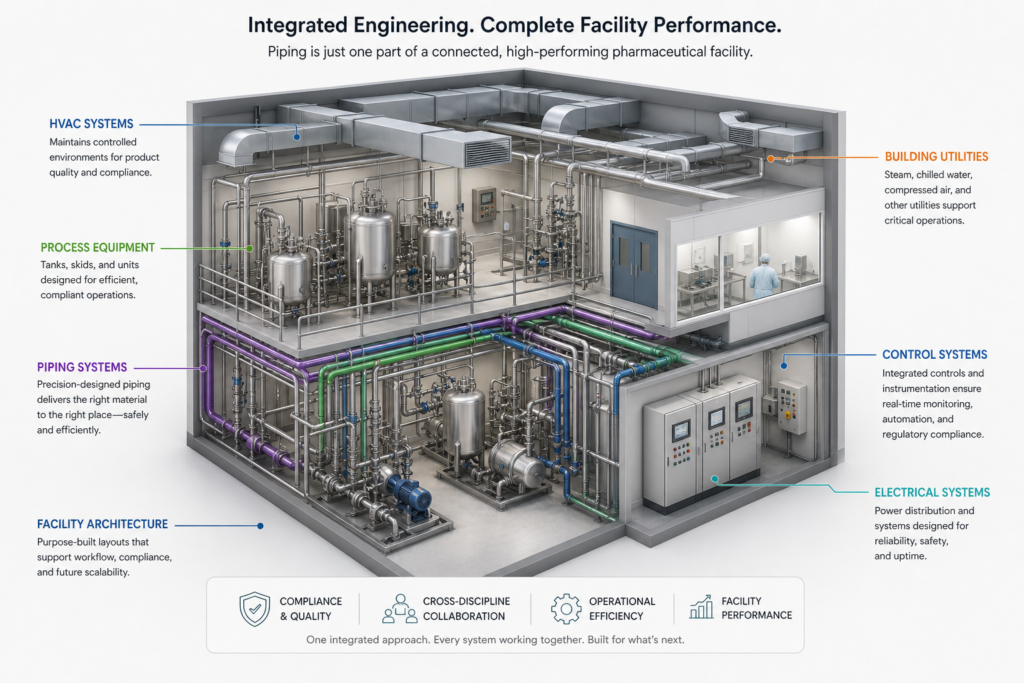
Piping design cannot be done in isolation.
It must be aligned with:
- Facility layout
- Equipment placement
- Utility systems
- Automation and control strategies
- CQV planning, safety requirements, and long-term maintenance needs
This is where integrated pharma engineering services create value.
By aligning all disciplines early, engineering teams can ensure that piping systems support both compliance and efficiency.
How Pharma Access Approaches Piping Design
At Pharma Access, piping design is treated as a critical component of overall facility performance.

Through advanced pharmaceutical engineering design and integrated execution, we ensure that pharmaceutical process piping systems are:
- Hygienic and compliant
- Efficient and reliable
- Easy to validate and maintain
- Constructible, inspection-ready, and aligned with GMP expectations
This approach minimizes risk and supports long-term operational success.
Conclusion
Process piping is often not the most glamorous component of a pharmaceutical facility. But it is arguably one of the most critical. From product quality to operating efficiency to regulatory compliance, piping has a hand in it all.
Done right, piping unlocks the ability of the facility to run safely and predictably, every time. In a business where the details can make or break you, process piping is one detail you don’t want to overlook.
FAQs
1. What is pharmaceutical process piping?
It refers to piping systems used to transport fluids such as water, steam, and process materials within pharmaceutical facilities.
2. Why is piping design important in pharma manufacturing?
It ensures hygiene, prevents contamination, and supports compliance with regulatory standards while enabling cleanability, maintainability, and reliable operation .
3. What are dead legs in piping systems?
Dead legs are sections of piping where fluid stagnates, increasing the risk of microbial growth.
4. What materials are used in pharmaceutical piping?
Materials like stainless steel are commonly used due to their corrosion resistance and hygienic properties, along with compatible valves, gaskets, elastomers, and fittings selected based on process and GMP requirements .
5. How does piping design impact validation?
Proper design ensures systems can be easily tested, cleaned, and validated, reducing delays during CQV.



